This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
Regular readers will know that salamanders have been covered on Tet Zoo on several occasions in the past, sometimes at great length and reasonable depth (see links below). Today I want to recycle a section of text that’s appeared here before, though what you see here is updated and augmented. It concerns one of the weirdest salamander groups of them all.

Yes, sirens (Sirenidae) are perhaps the strangest of salamanders. Indeed, they’re so strange that at times they’ve even been excluded from Caudata (the salamander group) and put on their own as a ‘new’ sort of amphibian group termed Trachystomata or Meantes. Fully aquatic and superficially eel-like (though able to move on land when necessary), they’re neotenic, lack a pelvis and hindlimbs, possess external gills, lack eyelids, are adept at burrowing in mud, and reach 95 cm in the largest species.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.

Sirens can move on land when required: this is Siren lacertina. Image in public domain, from here.
Sirens of the past and changes in body size. Having mentioned size, the extinct Cretaceous-Paleocene siren Habrosaurus reached 1.6 m, which is enormous and frightening. Habrosaurus, in fact, is so spectacular that I was half-expecting there to be some life restorations of it online (there are two species: H. prodilatus from the Campanian of Alberta, and H. dilatus from the Maastrichtian and Paleocene of Wyoming and Montana). Alas, that doesn’t seem to be the case, so I had to resort to creating this…
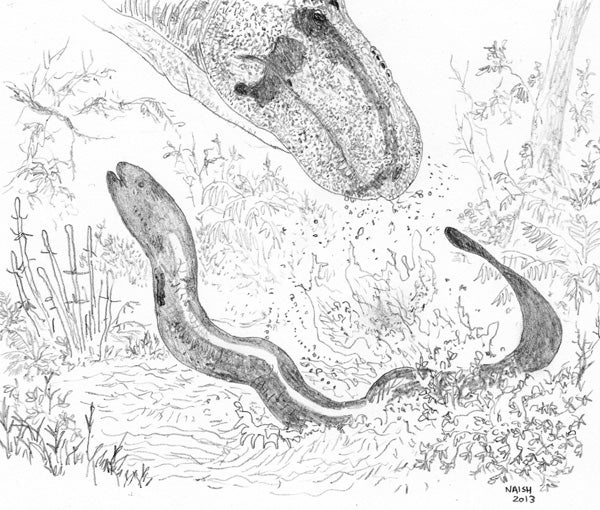
A scene from Late Cretaceous North America: a drinking tyrannosaur darts away in surprise as a panicking Habrosaurus bursts from the edge of shallow water in a flurry of spray. The habrosaur is a big individual, 1.6 m long. Illustration by Darren Naish.
The fact that Habrosaurus is so large raises the inevitable question about trends in body size: have sirens consistently gotten smaller since the Cretaceous? Bonett et al. (2013) examined body size variation across siren evolution and revealed a complex picture of ‘body size shuffling’. Large species gave way to smaller ones that evolved during the Paleocene-Eocene Thermal Maximum, but large species evolved again, later on. Interestingly, giant sirens evolved when giant sympatric amphiumas were already present (for more on amphiumas – another group of large, eel-like, aquatic, salamanders – see the links below).
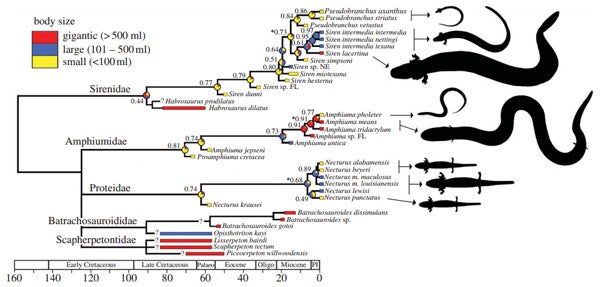
Time-calibrated salamander cladogram from Bonett et al. (2013), depicting actual and inferred body sizes for sirens, amphiumas and other North American groups. One interesting detail: Pseudobranchus appears to be nested within Siren.
Beak and teeth. Sexual dimorphism is obvious in at least some living siren species: males are larger overall and have proportionally bigger heads (Reinhard et al. 2013), a non-trivial fact that seems to be important as goes siren reproduction (read on). One of the biggest surprises about the group is that they have a horny beak and pavements of teeth on the palate. The beak forms a broad platform inside the jaws, and the jaw joint is ventrally displaced relative to the rest of the skull. All of these features are adaptations for crushing, and field studies show that sirens feed extensively on gastropods and bivalves... though they eat other things as well, on which read on...

Skull of Siren lacertina, with massive hyoid apparatus below and left forelimb visible in the background. You should be able to make out the hooked beak in the upper jaw, the beaks lining the lower jaws, and the suggestion of teeth in the upper jaw. Photo by HCA, from here, CC BY-SA 4.0.
Sirens are unlike most salamanders in that their teeth are (usually) not pedicellate: that is, their teeth are not connected to the jaw bones by way of a flexible pedicle at the tooth’s base. Salamanders start their life as larvae without pedicellate teeth and normally develop the condition as they mature, so lack of pedicelly is seen as a neotenous condition. Proteids (mudpuppies and olms) also lack pedicellate teeth as adults, as do the extinct batrachosauroidids and the Jurassic salamanders Kokartus and Beiyanerpeton. The fact that certain early salamanders (including stem members of the group) lack pedicellate teeth seems to challenge the hypothesis that pedicelly is a synapomorphy of Lissamphia and a primitive, inherited character for salamanders (Gao & Shubin 2012).

Captive Lesser siren (S. intermedia) in profile. Image by Stan Shebs, from here, CC BY-SA 3.0.
Courtship, mating, nest-building and nest guarding. Until recently, very little was known of siren biology and behaviour – we still don’t know all that much, but recent studies have shed light on several aspects of siren biology. Exactly how sirens reproduce has long been the source of some disagreement, and this is mostly because their biology seems contradictory. I need to remind you to begin with that salamanders of many groups practise internal fertilisation via the production of a specialised sperm capsule – termed a spermatophore – that’s picked up (cloacally of course) and absorbed by the female. A female siren is said to lay her egg clutch over a period of a few days (rather than all in one go), a system which obviously makes it look as if the eggs have been fertilised internally and, ergo, that spermatophores are produced by males. However, the male siren cloaca lacks any and all of the structures required for spermatophore production, so… what gives?

Select glimpses of siren reproductive behaviour. (a) Male Lesser siren guarding eggs in nest. (b) Male and female in nest together - note prominent sexual dimorphism. (c) Week-old hatchling. Images from Reinhard et al. (2013).
Reinhard et al. (2013) studied siren reproductive behaviour in the lab. A lot of neat stuff happened. Males were observed building nests: they selected a sheltered site, plucked moss, and then dragged it to the nest area. Other individuals were encouraged to stay away from the nest through biting. The idea of a nest-building salamander might seem odd, but it isn’t wholly radical since we’ve long known that other aquatic salamanders (like Asian giant salamanders and hellbenders) construct, maintain and guard breeding burrows.

Regular readers might recall this cartoon, depicting reproductive strategies practised by Asian giant salamanders. We now know that sirens do things somewhat similarly, the large males building nests and then guarding the eggs and larvae from other individuals. Image by Darren Naish.
Moving on, courtship occurred within the nest site, the male and female indulging in ritualised behaviours involving circling, pursuit, flank rubbing, tail undulation and head rubbing. This continued for about 2 hours, eventually culminating in the female turning onto her back and discharging approximately 120-130 tiny eggs into the nest substrate (a moss). The male then also inverted and fertilised the eggs. So, this proves it: sirens do indeed practise external fertilisation (Reinhard et al. 2013). The female then left, and the male stayed to guard the nest. He drove intruding individuals away, fanned the eggs with his tail, frequently moved the egg mass, and kept the nest area clean. Once the eggs hatched (after about a month), the male defended the larvae, even though both the male and the larvae left the nest on occasion before returning to it. This baby-guarding continued for about a week after hatching.
A selection of salamanders drawn for my textbook project... argh, without a siren! This and numerous other images visible here at the Tet Zoo patreon. Image by Darren Naish.
Sexual dimorphism explained? It looks likely that this nest-guarding and baby-guarding is linked to the sexual dimorphism mentioned earlier. Typically in salamanders, it’s the females who are larger, but what seems likely for sirens is that male size has evolved in step with nest guarding (Reinhard et al. 2013). Curiously, this isn’t the case in other salamanders where males are nest-guarders (Asian giant salamanders and hellbenders). Perhaps sirens are different in that their extremely high population densities mean that intraspecific predation on eggs and larvae needs to be counteracted in a more proactive way – sirens are surprisingly abundant at some localities, there being more than two individuals for every square metre in some ideal habitats.
Siren diet and feeding ecology is also remarkable: they are apparently partially herbivorous, ingesting vascular plants and algae on occasion. They have enlarged hindguts that may house symbiotic microbes (Pryor et al. 2006). I’ve written about siren herbivory in the past and there’s often been a bit of scepticism about it – after all, aquatic salamanders of many sorts are known to ingest plant parts on occasion, apparently by mistake, so are we really sure that the ‘herbivory’ reported for sirens is intentional and important?

Nice whole-body shot of captive Lesser siren (S. intermedia). Image by Stan Shebs, from here, CC BY-SA 3.0.
Hill et al. (2015) recently look specifically at this subject and confirmed that facultative herbivory in sirens is real, deliberate, well documented (albeit sometimes misinterpreted), and probably present throughout the group. They recorded captive sirens frequently eating boiled lettuce and got photos of them doing it. Obviously, this makes sirens unique among caudates. While we’re here, remember that there are plant-eating frogs too.

View of Siren lacertina from above, showing a nice view of the gills. Image in public domain, from here.
A few other things about sirens are unusual as well. They can survive desiccation by forming a sort of mucus cocoon in the mud, a habit similar to that better known for lungfishes. Oh yeah, and they emit a yelping noise when grabbed (Halliday & Verrell 1986).
For previous Tet Zoo coverage of salamanders, see...
Spiky-frilled, lek-breeding amphibious salamanders… or ‘newts’
Coprophagy and the giraffe-neck program: more on plethodontids
The USA is still yielding lots of new extant tetrapod species
Refs - -
Gao, K. & Shubin, N. H. 2012. Late Jurassic salamandroid from western Liaoning, China. Proceedings of the NationalAcademy of Sciences, USA 109, 5767-5772.
Halliday, T. R. & Verrell, P. 1986. Salamanders and newts. In Halliday, T. & Adler, A. (eds) Animals of the World: Reptiles and Amphibians. The Leisure Circle (Wembley, UK), pp. 18-29.
Hill, R. L., Mendelson, J. R. & Stabile, J. L. 2015. Direct observation and review of herbivory in Sirenidae (Amphibia: Caudata). Southeastern Naturalist 14, N5-N9.
Pryor, G. S., German, D. P. & Bjorndal, K. A. 2006. Gastrointestinal fermentation in greater sirens (Siren lacertina). Journal of Herpetology 40, 112-117.