This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
In this, the third and final article in this series, we look at the few taxa not discussed so far. I’ve left them until last for two reasons. One is that they include the most recently named members of the group. Two is that the places they occupy in chameleon phylogeny are particularly informative as goes what we think about the evolutionary history of these remarkable lizards. What am I getting at? Read on…
Palleon from Madagascar. We’ll begin with Palleon, named in 2013 (Glaw et al. 2013) for two distinctive Madagascan species (P. nasus and P. lolontany, the latter only named in 1995) previously included within Brookesia. They’re small (c 4-9 cm in total), short-tailed, laterally compressed in the body and with a wave-like dorsal crest and beak-like nasal appendage in males. In fact these chameleons are so distinctive that some studies have hypothesised that they might be the sister-group of all other chameleons (Raxworthy et al. 2002). More recent analyses have found them to be part of the Brookesia lineage, though outside the clade that contains all definite Brookesia species (Townsend et al. 2011, Tolley et al. 2013).

P. nasus: photo by Axel Strauß, CC BY-SA 3.0.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
The name Palleon is a contracted version of ‘Palae-leon’ and refers to the inferred ancient origin of this little group (Glaw et al. 2013). If Palleon-lineage chameleons really have been around for 50 or 60 million years or more (as suggested by molecular divergence data) you’d expect there to be more than two species: are they another of those lineages that’s been a chronic ‘slow evolver’ relative to their close kin? It seems so, though we should of course be on the lookout for additional member of the lineage.

Close-up of P. nasus. Check out those details of head ornamentation as well as the spines along the back. Photo by Heinonlein, CC BY-SA 4.0.
Anyway… the genetic and morphological support putting these animals close to Brookesia (Townsend et al. 2011, Glaw et al. 2013, Tolley et al. 2013) means that Brookesiinae currently contains both Palleon and Brookesia.
Archaius of the Seychelles. Next up is Archaius – a name used solely for the Seychelles tiger chameleon A. tigris. Yes, a chameleon endemic to the Seychelles – what gives? Archaius isn’t a new name (it was published by Gray in 1865), but it’s only recently been resurrected, having long been subsumed into Chamaeleo, and more recently into Calumma. The Tiger chameleon is small (total length = c 16 cm) and with a pointed projection on its chin. Its parietal crest (the one that runs sagitally along the upper surface of the braincase) is forked, as is also the case in some Trioceros species.

Archaius tigris, the Seychelles Tiger chameleon. Photo by Hans Stieglitz, CC BY-SA 3.0.
While Archaius might not be especially remarkable from a morphological point of view, the really interesting thing about it is where it sits in the phylogeny. For, despite looking enough like a ‘conventional’ chamaeleonine chameleon that it has often been regarded as one, molecular data indicates that Archaius is actually most closely related to Rieppeleon, and hence is outside the clade that includes Rhampholeon and remaining chamaeleonines (Townsend et al. 2011, Tolley et al. 2013). Why is this a big deal?
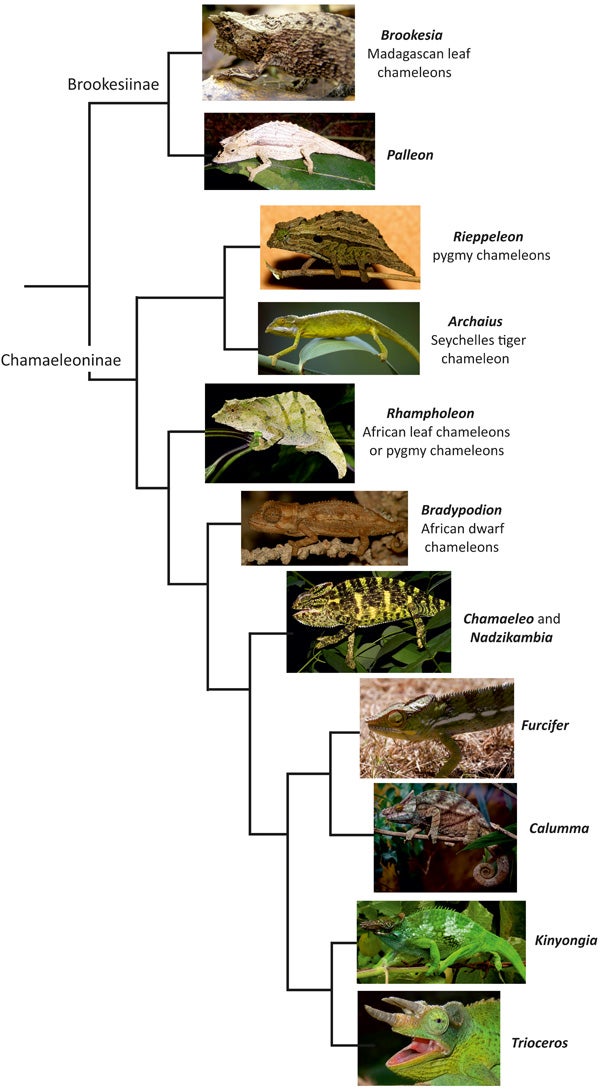
A more complex depiction of chameleon phylogeny (relative to versions shown in the previous two articles), based on Tolley et al. (2013). Brookesia by Frank Wouters, CC BY-SA 2.0; Palleon by Axel Strauß, CC BY-SA 3.0; Rieppeleon by R. J. Blach, CC BY-SA 3.0; Archaius by Hans Stieglitz, CC BY-SA 3.0; Rhampholeon by Martin Nielsen, CC BY-SA 4.0; Bradypodion by Javier Abalos Alvarez, CC BY-SA 2.0; Chamaeleo by Raju Kasambe, CC BY-SA 3.0; Furcifer by Gunnar M. Kvifte, CC-BY; Calumma by Steve Wilson, CC BY 2.0 Kinyongia by Ales Kocourek, CC BY 2.0; Trioceros by Benjamint444, CC BY-SA 3.0.
You’ll recall that Rieppeleon and Rhampholeon are both ‘leaf chameleons’: small, short-tailed, cryptic chameleons that lack some of the key anatomical and lifestyle features associated with ‘conventional’ chameleons. Furthermore, brookesiines are ‘leaf chameleons’ of a sort, as well. Look at the phylogeny, employ your Dawkinsian powers of concestor reconstruction, and it would look as if chameleons were all ‘leaf chameleons’ right up to the origin of the clade that includes Bradypodion, Chamaeleo and kin.
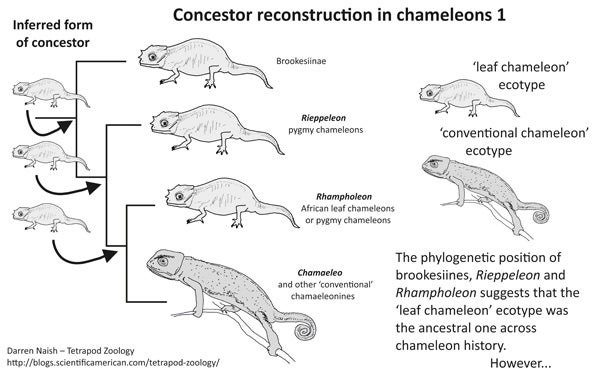
This tree-based hypothesis has some interesting ramifications. It would suggest that key chameleon traits evolved within the context of especially small size, a cryptic lifestyle and an ecology that mostly involved leaf litter, low herbage and twiggy shrubs. It would also mean that we might seek to explain why, at some point during the leafy, tropical Eocene, chameleons suddenly evolved larger size, prehensile tails, and morphed into (relatively) giant, flamboyant, ornamented beasts associated with tall trees.
Here is where Archaius saunters in and says “No... probably” to the above. Its position suggests that the ‘leaf chameleon’ way of doing things was not retained across chameleonhood until late in the game: the position of Archaius makes it at least as likely that the ‘conventional’ chameleon ecotype is the ancestral one, and that the ‘leaf chameleon’ ecotype is the weird, divergent one, at least within Chamaeleoninae. This would mean that the ‘leaf chameleon’ ecotype shared by Rieppeleon and Rhampholeon is convergent.
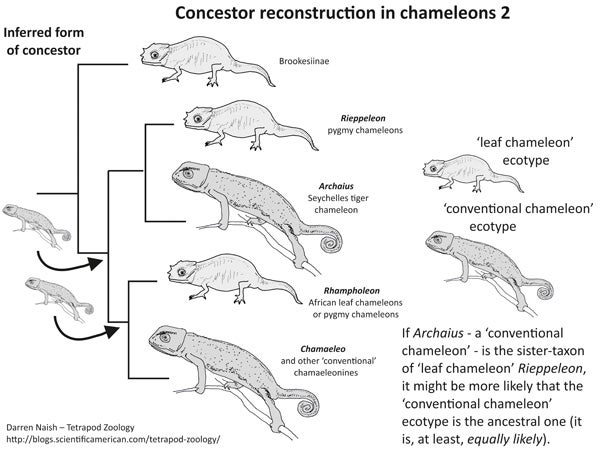
The presence of this species on the Seychelles is presumably the result of a single over-water dispersal event from Africa (Townsend et al. 2011), and likely not from Madagascar as argued by Raxworthy et al. (2002). In fact, chameleons as a whole – well, the crown-chameleon clade, anyway – seem to have originated on Africa and dispersed from here multiple times. Why this ‘eastwards from Africa’ hypothesis, rather than one invoking long-time residence on various broken fragments of Gondwana? The reason is that crown-chameleons aren’t old enough for Gondwanan fragmentation to explain their presence in such places as Madagascar or the Seychelles. Crown-chameleons might be 60 million years old or so, but a group would need to have originated more than 80 million years ago to get stranded on the Seychelles as a consequence of Gondwanan fragmentation (Raxworthy et al. 2002, Townsend et al. 2011).

Two of the most familiar of Kinyongia chameleons. Left: K. fischeri, photo by Denise Chan, CC BY-SA 2.0 [UPDATE: no, this is K. matschiei: see comment below by CVAnderson]. Right: K. xenorhina, photo by David Udvardy, CC BY-SA 3.0.
Kinyongia, kin of Trioceros. Next up is Kinyongia, an east African taxon named in 2006 for a group of 10 species previously included in Bradypodion (see the previous article for more on Bradypodion) (Tilbury et al. 2006). The number of species has about doubled since then. Kinyongia chameleons tend to be mid-sized animals of highland habitats in Kenya, Tanzania, the eastern Democratic Republic of the Congo and so on. They’re usually sexually dimorphic, the males possessing short, robust rostral protuberances. The most familiar of the lot is probably Fischer’s chameleon K. fischeri, a species sometimes said to have the longest tongue of any chameleon. I have no idea whether this is true or not [UPDATE: it's not. See comment below by CVAnderson].

Front cover of Spawls et al. (2004), featuring Kinyongia xenorhina.
Some Kinyongia species are especially odd. Among these is the Strange-nosed chameleon or Single welded-horn chameleon K. xenorhina, males of which are equipped with a laterally compressed, oval rostral projection that’s similar in size and shape to the casque. A great photo of one of these chameleons adorns the cover of Spawls et al.’s A Field Guide to the Reptiles of East Africa (Spawls et al. 2004). Then there’s the Uluguru one-horned chameleon or Sharp-nosed chameleon K. oxyrhina of the Tanzanian Uluguru and Udzungwa Mountains, males of which have a long, blade-like horn. And there are others…
You might have noted that these animals sound Trioceros-like overall (Trioceros being the mostly Afromontane chameleon group that famously includes several horned species). Indeed, some molecular phylogenetic studies find the two to be sister-groups (Tolley et al. 2013).

K. fischeri in Tanzania, most familiar of the Kinyongia chameleons [UPDATE: no, this is K. multituberculata: see comment below by CVAnderson]. Photo by Jjgodox, CC BY-SA 4.0.
Nadzikambia. Finally, we come to another recently named taxon: Nadzikambia, named in 2006 in the same study that also named Kinyongia (Tilbury et al. 2006). Only two Nadzikambia species are currently known: the Mlanje Mountain chameleon N. mlanjensis from Malawi, named in 1965, and the Mount Mabu chameleon N. baylissi from Mozambique, named in 2010. What appears to be a third species was reported (but is as yet unnamed, so far as I can tell) in 2014. All are fairly conservative, mid-sized chameleons with a low casque and finely heterogenous scalation (Tilbury et al. 2006). N. baylissi is famous because its discovery (by Julian Bayliss) resulted from a targeted search involving the identification of the right kind of forest via Google Earth (for more on this amazing story, go here).

Juvenile N. baylissi: a screengrab from a short film available here.
It’s always been thought that Nadzikambia is close to, or even part of, Chamaeleo. And some recent molecular studies finds the two to be sister-taxa (Tilbury & Tolley 2009, Tolley et al. 2013), the two perhaps diverging at the start of the Oligocene, about 30 million years ago (Tolley et al. 2013). Another study (Townsend et al. 2011) found Nadzikambia to be closest to Bradypodion, the inferred divergence date of the two being put at over 40 million years ago. At the risk of repeating myself, consider these inferred divergence dates relative to the divergence dates of other, more familiar tetrapod groups. These rather similar chameleon lineages have been distinct for longer than deer have been distinct from antelopes and cattle, or walruses have been distinct from seals.
There aren't any Nadzikambia images online that are widely available for use, so here's another picture of a Kinyongia: this is K. vosseleri (based on a photo by Jan Stipala). I initially prepared this illustration because I thought it was a depiction of Nadzikambia, sigh...
Maybe there are two ways of looking at this. One is to say that chameleons are fairly conservative, relatively little morphological and ecological change having accrued in the timeframe concerned. Another is to say that the lineages are really far more disparate, ‘more distinct’ than we’ve conventionally assumed, and that we’ve understated how much evolution and diversification might actually have evolved.
That’s it, for now. And with that, we’ve done it: looked briefly at all major, named lineages within the crown-chameleon radiation. There is, of course, so much more to say. Much could be said of chameleon anatomy and functional morphology, of the fossil history and broader affinities of chameleons within the lizard clade Iguania, and of chameleon ecology, behaviour and lifestyle. Plus there are an enormous number of species – a great many of them recently named – that really warrant a more detailed look. But, for now, this will do. I hope you’ve enjoyed this look at chameleon diversity and I’m sure we’ll be returning to them in time.
For previous Tet Zoo articles on iguanian lizards, see...
Harduns and toad-heads; a tale of arenicoly and over-looked convergence
The Squamozoic actually happened (kind of): giant herbivorous lizards in the Paleogene
Australia, land of dragons (by which I mean: agamids) (part I)
By the Horns of Trioceros, the Casque of Calumma, the Brood of Bradypodion--Chameleons, Part 2
Refs - -
Glaw, F., Hawlitschek, O. & Ruthensteiner, B. 2013. A new genus name for an ancient Malagasy chameleon clade and a PDF-embedded 3D model of its skeleton. Salamandra 49, 237-238.
Raxworthy, C. J., Forstner, M. R. J. & Nussbaum, R. A. 2002. Chameleon radiation by oceanic dispersal. Nature 415, 784-787.
Tilbury, C. R. & Tolley, K. A. 2009. A re-appraisal of the systematics of the African genus Chamaeleo (Reptilia: Chamaeleonidae). Zootaxa 2079, 57-68.
Tilbury, C. R., Tolley, K. A. & Branch, W. R. 2006. A review of the systematics of the genus Bradypodion (Sauria: Chamaeleonidae), with the description of two new genera. Zootaxa 1363, 23-38.
Tolley, K. A., Townsend, T. M. & Vences, M. 2013. Large-scale phylogeny of chameleons suggests African origins and Eocene diversification. Proceedings of the Royal Society of London B, 280, 20130184.
Townsend, T.M., Tolley, K. A., Glaw, F., Böhme, W. & Vences, M. 2011. Eastward from Africa: palaeocurrent-mediated chameleon dispersal to the Seychelles islands. Biology Letters 7, 225-228.