This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
Mention the term ‘mole-rat’ (or molerat, or mole rat) and most people (zoologists included) will think of Heterocephalus glaber, the remarkable Naked mole-rat or Sandpuppy of Ethiopia, Somalia and Kenya. But this is merely one of a whole radiation of African mole-rats, the species of which occur right across sub-Saharan Africa. Given that there’s an absolute crap-ton of information online about the Naked mole-rat, I’m mostly going to avoid discussing that species here [image used at top shows Ansell's mole-rat Fukomys anselli; photo by Headster, CC BY 3.0].

A Naked mole-rat queen - either cute and nice to look at, or the grotesque stuff of nightmares. Image by Chomez, in public domain.
The first thing we have to say is that we’re talking specifically here about bathyergid mole-rats or blesmols, and bathyergid mole-rats or blesmols alone: other morphologically and ecologically similar rodent species are also called mole-rats, but they’re not part of Bathyergidae. Those species – they include the Eurasian blind mole-rats of the genera Nannospalax and Spalax – are included within a group termed Spalacinae or Spalacidae. They appear to be muroids – part of the enormous rodent group that includes mice, rats, hamsters, voles, gerbils and so on – and hence are not even close to bathyergids.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
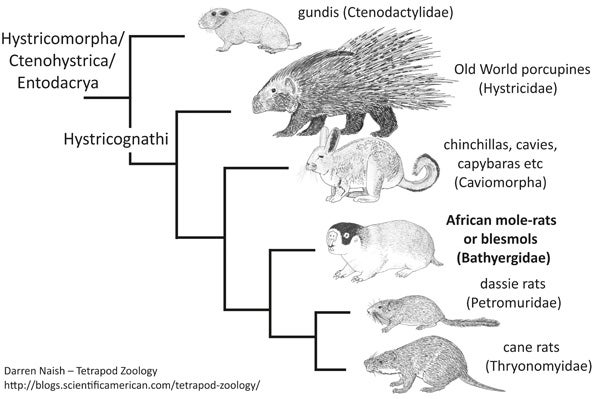
Hystricomorph/ctenohystrican/entodacryan cladogram, based on the molecular phylogenies published by Huchon et al. (2002) and Fabre et al. (2012). This tree (and several other rodent cladograms) are from my in-prep textbook.
If you’re wondering, bathyergids appear to be part of Hystricomorpha or Ctenohystrica, the gundi-porcupine-caviomorph clade also known as Entodacrya. Within this clade, bathyergids appear to be close to dassie rats (Petromuridae) and cane rats (Thryonomyidae), and then to Old World porcupines and caviomorphs (Huchon & Douzery 2001, Huchon et al. 2002, Fabre et al. 2012, Patterson & Upham 2014). Some (but not all) of these groups have conventionally been included within a hystricomorph assemblage termed Phiomorpha. There’s more to say than that as goes phylogeny and taxonomy, of course, but we must move on.

Hystricomorph/ctenohystrican/entodacryan rodent montage. The groups shown here are cane rats, dassie rats, gundis, Old World porcupines, African mole-rats and caviomorphs (represented by the chinchillid Lagidium). Images from the Tet Zoo textbook project.
Bathyergid mole-rats are compact, short-tailed rodents with tiny eyes and near-invisible ears. Their large white incisors are exposed even when the mouth is closed, the lips actually closing behind the incisors. Prominent grooves are present on the incisors of some species. The incisors in the lower jaw are able to move independently (is this unique among mammals?), their long roots occupying a huge portion of the skull’s interior and adding to its great strength (“a steel rod, a square millimetre in section, only penetrated when a weight of 3 kg was applied – this is equivalent to about 8cwt per sq in” (Hanney 1977)). Long claws are typical, as is very short, velvety fur of the sort present widely in subterranean mammals (Heterocephalus being the obvious exception).
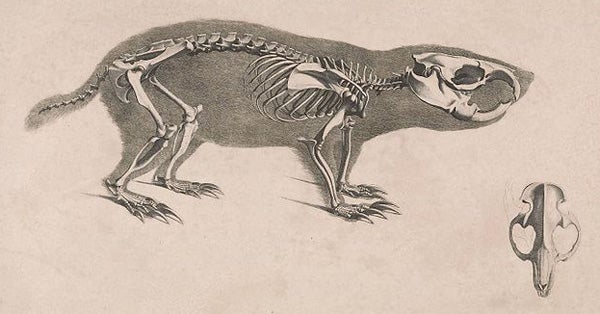
The skeleton and skull of Bathyergus suillus, as illustrated by Christian Heinrich Pander in 1821 (image in public domain). I mostly see claws and incisors.
Bold markings are present in some species, with dark masks, pale eye-rings and prominent dichromatism being obvious in Georychus in particular. I wonder why this is – it isn’t what you’d expect for a subterranean group that are hardly able to see anything. Mole-rats are said to make “interesting pets”, but that (surprise surprise) “some species bite unexpectedly; even the newborn can inflict severe bites” (Nowak 1999, p. 1636).

The rather boldly marked Cape mole-rat or blesmol (Georychus capensis) in life. Photo by Nigel C. Bennett, CC BY-SA 3.0.
Mole-rats are herbivorous, and most species rely on geophytes (storage organs) and roots, though grasses, herbs and occasionally invertebrates and even other rodents are eaten by some species too. Several species store plant parts in special burrow chambers and some will only eat part of a root or geophyte, apparently deliberately leaving it to recover and regenerate such that it can be exploited over the long term. The burrows they construct can be absurdly long: people have discovered common mole-rat burrow systems exceeding 1 km in length, and some Naked mole-rat burrows are over 3 km long (Jarvis 2001). Not a typo: over 3 km.

A rather angry-looking Fukomys anselli. Photo by Anfimo, in public domain.
None of the several genera are especially familiar or well known. In addition to Heterocephalus, there’s Cryptomys (containing the seven or so species of common mole-rat), Fukomys (named for a set of species included within Cryptomys until 2006), Georychus (the Cape mole-rat), Heliophobius (the Silky mole-rat), and Bathyergus (the dune mole-rats). Some of the common mole-rat species are colonial (some others are solitary), the non-breeding individuals collecting roots, excavating tunnels, and caring for the young of the single breeding pair. This mention of coloniality and a single breeding pair will of course remind you of the eusocial system present in Naked mole-rats, whereby an especially big Queen is the lone breeding female in the relatively enormous colony, the individuals of lower castes having a suppressed reproductive ability and living a life where all they do is work: they maintain the burrows, care for the babies and so on. And, yes, the colonies are huge relative to what you might guess: they average 80 individuals, and can consist of as many as 300.

Somewhat surreal image of a captive Fukomys mechowi. Photo by Nikolai Barbel, CC BY-SA 3.0.
For a while the general assumption seemed to be that the eusociality of the Naked mole-rat had evolved from the also eusocial but less ‘extreme’ biology of the Cryptomys and Fukomys common mole-rats… or, at least, evolved from common-mole-rat-like mole-rats. These eusocial mole-rats (the eusocial common mole-rats and the Naked mole-rat) inhabit more arid places than most other mole-rats, and they’re the most specialised when it comes to reliance on geophytes and roots, so the idea of some sort of trend toward arid-land specialisation, ever-smaller size, geophyte/root specialisation and increasing eusociality looked like a possibility.
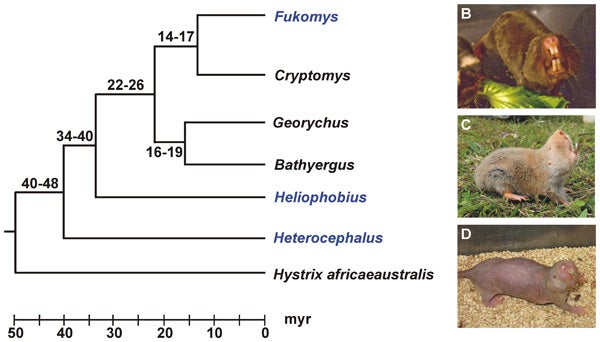
At left, a molecular phylogeny of bathyergid mole-rats - note how old the Heterocephalus divergence is, and how Heterocephalus is the sister-taxon to the rest of the clade. The images at right show (B) Fukomys, (C) Heliophobius, and (D) Heterocephalus. B and C by Sharry Goldman; D by Radim Šumbera. Entire image from Seney et al. (2009), CC.
But this never made sense given that the Naked mole-rat is not nested within Cryptomys or Fukomys. Instead (look at the molecular phylogeny above, from Seney et al. (2009)), it seems to be the oldest-diverging lineage within the group, molecular clock data positing its divergence at something like 40 million years ago (a possibility as yet unsupported by fossils, incidentally: there are quite a few fossils of Heterocephalus – three extinct species have been named, no less – but the oldest of them are from the Miocene). Maybe, then, the eusociality present in common mole-rats and Heterocephalus evolved convergently? By mapping patterns of social behaviour onto a molecular phylogeny, Faulkes et al. (2004) hinted at the possibility that eusociality might even be the primitive condition for Bathyergidae, and that solitary lifestyles have evolved from this condition. This seems rather radical, especially given what a big deal eusociality is (it’s a strategy otherwise unknown in mammals), but at least it’s plausible.
Heterocephalus is so distinct relative to all other members of the bathyergid lineage, by the way, that Patterson & Upham (2014) argued that it should be given its own ‘family’: Heterocephalidae. You can see their logic, even if (like me) you regard monotaxic ‘families’ like this as philosophically redundant (by which I mean that the Heterocephalus lineage already has a unique moniker, this being Heterocephalus).
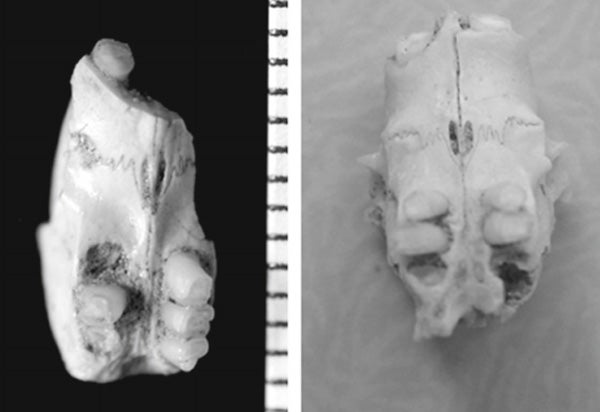
There are a lot of fossils of Heterocephalus, the lineage that included the Naked mole-rat. These partial skulls - both from the Pliocene of Tanzania - represent the extinct species H. manthii (at left) and H. quenstedti (from Denys 2011).
Dune mole-rats (Bathyergus) also live in groups (these consisting of around five individuals). They’re unusual in being restricted to the arid, sandy south-western corner of Africa, and in being especially big: big males of the Cape dune mole-rat B. suillus can reach 33 cm in length and 933 g in mass, making this species the biggest living subterranean rodent.
Why are bathyergids restricted to sub-Saharan Africa, if they’re so ancient? They really should be more widespread, and we have tantalising indications that they were: a Lower Miocene mole-rat has been reported from Israel, though it has yet to be described in detail (to my knowledge). Kingdon (1997) suggested that their absence from northern Africa is due to their having been replaced by a younger group with a similar ecology, the root-rats or rhizomyids, while Faulkes et al. (2004) proposed that the spalacine Spalax might have been the ‘replacer’. Maybe both of those groups usurped bathyergids. Alas, as always with proposals of this sort, they’re not especially satisfying since we have no real data showing that either root-rats or spalacines are in any way ‘superior’ to bathyergid mole-rats, nor (at the moment) is it clear why competitive replacement would occur and lead to the extinction of the group.

Did members of either of these two subterranean rodent lineages - Tachyoryctes (at left) and/or Spalax - somehow displace bathyergid mole-rats from areas where they once occurred? Tachyoryctes image by Eduard Rüppell, in public domain (I can't believe there are no good Tachyoryctes photos in the public domain). Spalax image by Vivan755, CC BY-SA 3.0.
Subterranean rodents are not especially familiar animals to the majority of we humans, but their diversity, widespread distribution and geological longevity show that they represent a hugely successful evolutionary event. And they harbour an amazing diversity of behaviours, social strategies, lifestyles, physiological regimes and anatomical adaptations. Furthermore, evolutionary processes have – as hinted at in the article here – repeatedly invented similarly adapted rodents again and again and again, in different lineages, and on different continents. We will visit those other subterranean rodents at some point in the future, oh yes. And I didn’t even mention mole-rat jaw musculature (apologies to Philip Cox).
And if you like rodents – and you clearly do, since you read the whole of this article – you really should buy this t-shirt, and maybe wear it too…

Available here. It’s the best selling Tet Zoo t-shirt!
For previous Tet Zoo articles on rodents, see…
Refs - -
Denys, C. 2011. Rodents. In Harrison, T. (ed) Paleontology and Geology of Laetoli: Human Evolution in Context. Volume 2: Fossil Hominins and the Associated Fauna. Springer, pp. 15-53.
Faulkes, C. G., Verheyen, E., Verheyen, W., Jarvis, J. U. M. & Bennett, C. 2004. Phylogeography patterns of genetic divergence and speciation in African mole rats (Family: Bathyergidae). Molecular Ecology 13, 613-629.
Huchon, D. & Douzery, E. J. P. 2001. From the Old World to the New World: a molecular chronicle of the phylogeny and biogeography of hystricognath rodents. Molecular Phylogenetics and Evolution 20, 238-251.
Huchon, D., Madsen, O., Sibbald, M. J. J. B., Ament, K., Stanhope, M. J., Catzeflis, F., de Jong, W. W. & Douzery, E. J. P. 2002. Rodent phylogeny and a timescale for the evolution of Glires: evidence from an extensive taxon sampling using three nuclear genes. Molecular Biology and Evolution 19, 1053-1065.
Kingdon, J. 1997. The Kingdon Field Guide to African Mammals. Academic Press, San Diego.
Patterson, B. D. & Upham, N. S. 2014. A newly recognized family from the Horn of Africa, the Heterocephalidae (Rodentia: Ctenohystrica). Zoological Journal of the Linnean Society 172, 942-963.