This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
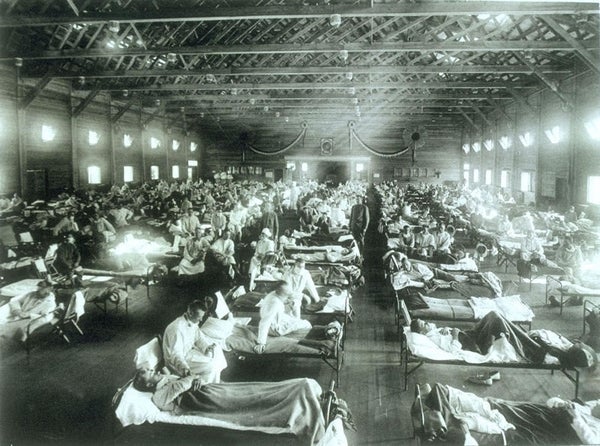
In 2018 the world will mark the 100th anniversary of the most devastating infectious disease event in recorded history: the 1918 influenza pandemic.
The severity of the event, which caused an estimated 50 million to 100 million deaths worldwide, likely resulted from several factors: First, most of the global population probably had no preexisting immune protection to the brand-new virus that had emerged. Evidence from the study of viral genes suggests the 1918 flu virus originated from an avian source and then somehow adapted to circulate efficiently in humans. Second, inherent properties of this virus may have contributed to its pathogenicity—the mechanisms by which it caused illness. Finally, efforts to control the spread of the pandemic and prevent morbidity and mortality were limited by conditions and technologies of the time. Crowding and poor sanitation allowed for rampant disease transmission, especially in areas where access to health care was limited. Antivirals to treat influenza were not available in 1918, and infections often were complicated by fatal bacterial pneumonias for which there were no effective antibiotics.
Further, protective vaccines—the cornerstone of modern influenza prevention—were still decades in the future. And even though we now have such vaccines, they likely would not be effective against a 1918-like virus. One hundred years later, we must reexamine our approach to influenza vaccines so we can avert a repeat of that worldwide catastrophe.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Over the past 100 years, apart from vaccines, substantial advances have been made in public health and health care infrastructure, particularly intensive care capacity. In addition, we now have antiviral drugs for influenza, antibiotics for secondary bacterial infections and influenza vaccines. Despite these developments, however, seasonal influenza epidemics still cause some 12,000 to 56,000 deaths each year in the U.S. These seasonal influenza viruses continually evolve by accumulating mutations in the genes that code for surface proteins and/or swapping genes with other influenza viruses. When the result is a small or modest change in the virus, it is referred to as an antigenic drift. We attempt to mitigate the burden of influenza by developing new seasonal vaccines for both hemispheres each year that target the influenza viruses surveillance suggests will be most common in the upcoming influenza season. Even though these vaccines are updated frequently, they may be only 40 to 60 percent effective against seasonal influenza viruses. And in some years vaccine–virus mismatches occur because new variants appear and spread in the interval between vaccine formulation and deployment. In these cases, vaccine effectiveness is considerably lower.
Importantly, seasonal influenza vaccines may provide little or no protection against pandemic influenza viruses where the new virus is considerably different from circulating seasonal viruses. Pandemic viruses typically arise from a process referred to as an antigenic shift, in which the new virus acquires, usually from animal influenza viruses, one or more genes that are entirely novel (as seems to have happened in 1918, when all eight pandemic virus genes were novel).
After 1918 three influenza pandemics associated with antigenic shifts occurred: in 1957, 1968 and 2009. In each of these instances, however, the new viruses emerged via the mixing or reassortment of animal influenza virus genes with those of the 1918-descended viruses already circulating in the human population. Likely explanations for the lesser severity of the last three pandemics compared with that of 1918 include lower viral pathogenicity, partial preexisting immunity to the new viruses and improvements in public health infrastructure and medical treatment. Vaccines were not a major factor, as effective vaccines were not available until well after the peak of each pandemic. The three recent influenza pandemics remind us that new pandemics are inevitable, and that current seasonal vaccines are unlikely to prevent or significantly control them.
We also must address the issue of “pre-pandemic” influenza viruses, that is, those that threaten to emerge to cause pandemics but that have not (yet) done so. In this regard, human infections with avian influenza viruses, such as Asian lineage A(H5N1) and A(H7N9), have occurred with increasing frequency over the past two decades. The risk of human adaptation of these viruses to cause a future pandemic remains unknown but vigilance is called for. Pre-pandemic vaccines against various strains of H5N1 and H7N9 viruses have been developed and in some cases stockpiled; similar to seasonal influenza viruses, however, these avian strains are subject to antigenic drift within their avian hosts. For example, many of the H7N9 avian viruses that have jumped species from poultry to cause human infections in China in 2017 have changed significantly from 2013 avian strains. As a result, the human immune responses elicited by a vaccine developed against the 2013 A(H7N9) virus may not be effective against its 2017 strains.
The remarkable capacity of influenza viruses to undergo antigenic drift or shift to overcome and escape human population immunity leaves us vulnerable to a public health disaster potentially as serious as the 1918 pandemic. To meet this global health challenge, scientists are working to develop “universal influenza vaccines”—new types of inoculations that can provide protection not only against changing seasonal influenza viruses but also against the inevitable pandemic viruses that will emerge in the future.
Recently the National Institute of Allergy and Infectious Diseases (NIAID) at the U.S. National Institutes of Health convened a scientific agenda-setting workshop with leading experts in the influenza field to address the need for better influenza vaccines. Among many hurdles to developing a universal vaccine, the most formidable is our incomplete understanding of the immune responses that protect people against influenza, including the role of immunity at mucosal surfaces.
Current influenza vaccines generate an immune response focused disproportionately on viral surface proteins called hemagglutinins. This response is mediated by antibodies, Y-shaped proteins made by the immune system to neutralize pathogens. Each influenza virus has one of the 18 different known hemagglutinin subtypes, and an antibody response to a vaccine or natural infection is specific to that subtype, leaving individuals vulnerable to attack by viruses containing hemagglutinins against which they have previously not been vaccinated for or infected by. Also, influenza virus hemagglutinins can mutate over time so that previously made antibodies no longer recognize them.
One approach to a universal influenza vaccine under active consideration is to design them to generate antibody responses to parts of the virus that are common to all influenza virus strains and do not readily change by mutation. One such component of influenza that remains relatively constant from virus to virus is a specific region of the hemagglutinin protein called the stalk, or stem. It is also important to clarify how other parts of the immune system such as immune cells work together with antibodies to protect against influenza. Although hurdles in the development of such universal vaccines are daunting, we are optimistic that we can apply existing tools and experimental approaches to meet the challenge.
As we approach the centennial of the 1918 influenza pandemic, let us be reminded of the importance of this line of research in preventing a repeat of one of the most disastrous events in the history of global health.