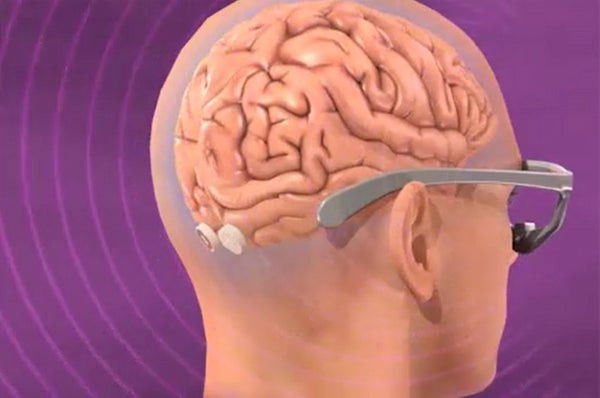
This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
In normal vision, light falls on the retinas inside the eyes, and is immediately transduced into electrochemical signals before being uploaded to the brain through the optic nerves. So you do not see light itself, but the brain's interpretation of electrochemical signals in the visual parts of the brain. It follows that, if your eyes do not work, but your brain is stimulated just so, your visual neurons will activate (and you will be able to see) just the same as if your eyes were in perfect condition.
Sounds easy, but can we do that? Building on decades of research in visual neuroscience, my lab, in collaboration with Susana Martinez-Conde’s, has now conducted some of the studies that validate this idea, completing some of the most important preliminary steps towards a new kind of visual prosthetic.
Francis Collins, the Director of the National Institutes of Health, has just posted a blog that highlights our approach. He took notice of our work when we first presented it at this year's meeting for the Principal Investigators of the BRAIN Initiative—the NIH led government funding initiative meant to spur research along on topics like brain implants. The BRAIN Initiative funds several agencies including the NIH, including the National Science Foundation, who kindly funded the grant driving our research thus far.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Our starting premise is that vision is fundamentally a thumbnail sketch. Even if 99.9% of your retina works fine, but the central 1/1000th of your visual field is broken, you will be legally blind. That central 0.1% of your visual field is about the same size as your thumbnail held up at arm's length. Because that central 0.1% of the retina is the visual sweet spot, it is the place where the visual magic happens. In fact, much of the remaining 99.9% of the retina’s main job is to help you detect where to move your eyes next. This means that we need to restore central vision in the blind, or we are not really restoring functional vision at all.
In addition, we also need to achieve prosthetic vision with the same acuity as normal vision, or patients will still be visually impaired. How good is good enough? The answer is in the stars. Go outside on a clear night, look up, and find the stars in the sky. Seems counterintuitive, but the biggest and the smallest-looking stars in the sky are actually the same size from the point of view of your retinal photoreceptors. Because all stars are so far away, the light that reaches Earth from each of them and falls on your retina subtends an area smaller than that of a single photoreceptor. But because of scatter in the atmosphere (and especially in your eye), brighter stars activate more photoreceptors, and therefore seem bigger than dimmer stars. Therefore, the dimmest star in the sky is the smallest thing you can possibly see. If we can prosthetically restore vision of objects of that size, then we can restore vision of any objects in the world—which for many patients will be life-changing and worth risking brain surgery.
Our recent research shows that it is possible to stimulate the visual brain at a specific point with enough precision to prosthetically evoke a single star in the sky, at maximal contrast, without stimulating unwanted neurons. That is what it means to stimulate the brain—just so—and restore the building blocks of vision.
There are two main steps to our project:
First, to stimulate a stage of visual processing with synaptic precision, just as the natural visual system does it, we need a way to activate neurons in the visual system prosthetically. We do this by injecting a custom virus into the region of the brain where the optic nerves first connect within the center of the brain. The virus injects genes into those cells and changes them into photoreceptors: now they are excitable by light. Because these neurons reach all the way to the visual cortex at the very back of the brain, and because they are laid out in a map that matches the visual world, we can stimulate their synaptic connections precisely with a video projector to recreate how they would react to real incoming visual inputs. We then target the central visual neurons and ultimately restore vision. That is the essence of our planned visual prosthetic.
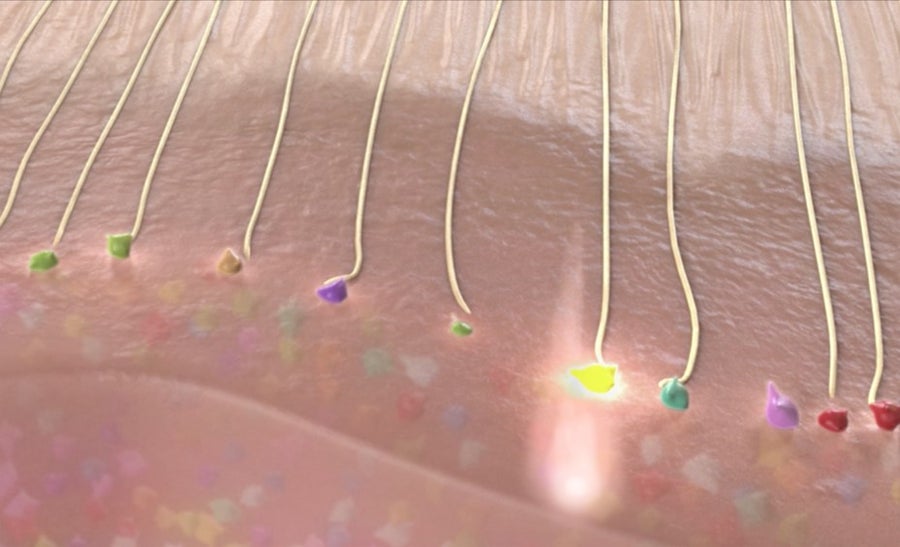
Activating inputs to visual cortex. Credits: Axoneme Studios, Courtesy of Stephen Macknik and Susana Martinez-Conde
Second, stimulating neurons without monitoring the effects of the stimulation will not be sufficient. We will also need to read-out the effects of our stimulation in real time, to make power adjustments as a function of the variability inherent to neuronal firing. To restore vision means to account for the regulatory mechanisms that keep the visual system calibrated at all times.
To accomplish this, we will inject more virus, now in the visual cortex, to make those neurons bioluminescent, so that they blink when they are active. By using many differently colored bioluminescent proteins, the neurons will have all sorts of different colors, meaning that we will be able to read-out their activity with a color camera—just like the tiny one on your cell phone.
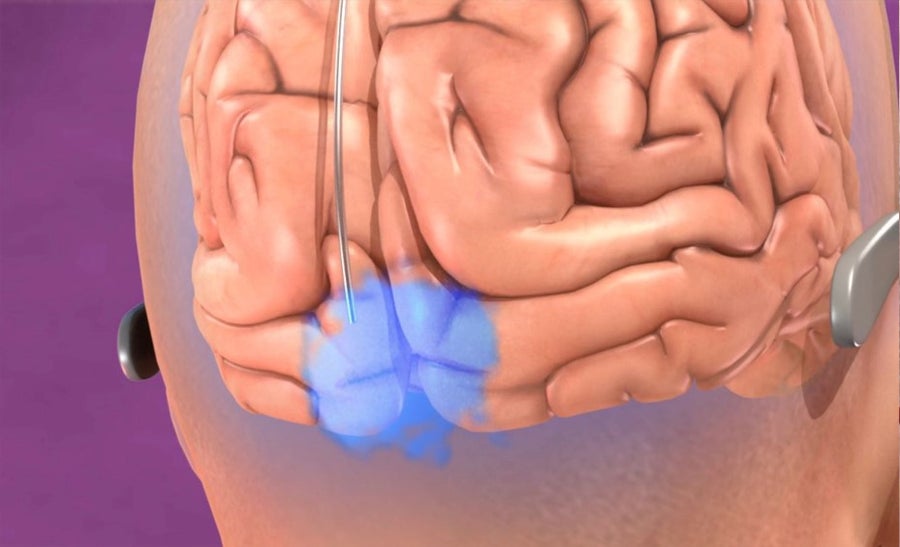
Bioluminescent viral delivery to visual cortex. Credits: Axoneme Studios, Courtesy of Stephen Macknik and Susana Martinez-Conde
Third, we will need to build a video projector/color camera that can be implanted in the brain. This is a relatively straightforward engineering task, comparable to or simpler than designing a smart phone. The next several years will reveal whether we can capitalize on these advances, to go from understanding the fundamental building blocks of prosthetic vision to making a functional high-quality visual prosthetic. We will keep you apprised of our progress along the way.