This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
As so much attention gets paid to the human microbiome and its impacts on human health, it can be easy to forget that plenty of other animals have microbiomes too. At the same time, it’s becoming more an more clear that we must consider the health of animals and yes even pests, if we hope to get a handle on infectious disease.
Mosquitoes have been in the news a lot lately due to the recent emergence of Zika virus a mosquito-borne infection that’s been linked to microcephaly in South America. Though the risk of Zika to pregnant mothers is grabbing headlines, mosquito-borne infections are nothing new. From malaria tochikungunya, pathogens transmitted by these insects plague the developing world. Malaria alone infects nearly 10% of the global population and causes over 2 million deaths every year.
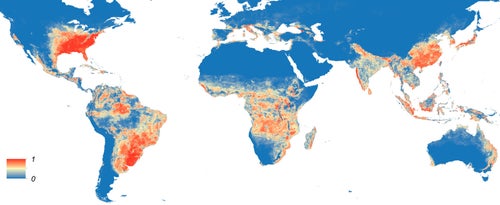
Map of mosquito prevalence. Source: http://elifesciences.org/content/4/e08347v3/figure2
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
All this is to say that understanding the biology of mosquitoes and other insects is important, and it’s becoming increasingly clear that the biology of an organism is tied to the biology of the microbes that colonize it.
Mosquito C-type lectins maintain gut microbiome homeostasis
This is hardly the first research that’s paid attention to the microbiology of mosquitoes, but most of the research I’ve seen focuses on mosquioto pathogens - the goal seems to be to alter mosquito behavior or outright kill them. However, the fact that human pathogens transmitted by mosquitoes pass through the insect gut, understanding how the normal microbiota is shaped could provide insights that will help curb these plagues (that’s assuming we don’t just wipe out the mosquitoes in the meantime).
These authors chose to focus C-type lectins (CTLs), a giant family of related proteins that bind to different types of carbohydrates found on the outside of cells. Lectins have a diverse range of functions, but many are involved in immunity, so it’s not a stretch to think they might be important in interacting with the bacteria in the mosquito gut. In mammals, CTLs bind to the cell walls of bacteria and help other cells to target them, or they trigger anenzymatic cascade that pokes holes in the bacteria’s cell membrane. CTLs have also been shown to help defend insects against bacterial infection.
But when Xiaojing Pang and co-authors eliminated expression of many different lectins, they saw something surprising (at least I was surprised) - the quantity of many normal commensal microbes actually decreased. In other words the production of these proteins, normally thought to aid in bacterial clearance, are somehow helping some bacteria to survive.
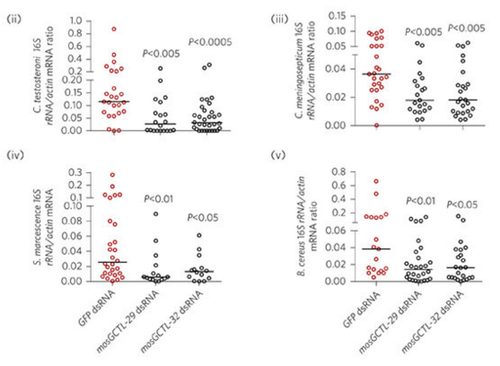
The populations of many normal gut microbes in mosquitoes actually decrease when certain C-type lectins are eliminated, suggesting that the CTLs normally help these bacteria survive. Source: Figure 2 of doi:10.1038/nmicrobiol.2016.2
It turns out, some CTLs protect the bacteria from a different arm of the mosquito immune response - antimicrobial peptides (AMPs). These smallish molecules are secreted into the gut (again of both insects and mammals) and typically grab onto and kill microbes that wander too close to the walls of the gut. But Pang et. al. show that the mosquito lectins are able to interfere with the binding of AMPs, thus protecting the good commensals bacteria from death.
The authors presume that this is a way of choosing which microbes live and which ones die. In order for this to work, the AMPs must be less specific than the CTLs that block them (otherwise, what’s the point of secreting the AMPs at all?). It’s a bit strange - both types of molecules are turned on by the same signals, and I’m a bit skeptical of the ability of these lectins to discriminate between friend and foe solely on the basis of what sugars they have on their cell surface - it’s unclear why commensals would have the sugar and pathogens wouldn’t.
And really, though the authors add the obligatory “this will help stop insect borne pathogens” at the end of the article (and though I tried to trick you to get you interested by introducing this post the same way), I’m not really sure this has any direct bearing on human disease. Then again, we know so little about this area of biology, I’m all for pushing the curtain back a little bit.