This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
In the thick black muck under a bay in the Philippines lives a creature the size a baseball bat, encased in a hard white shell. For hundreds of years, it somehow escaped formal description and dissection. Its name is the giant shipworm, and scientists from the United States and the Philippines have at long last cracked a live one open.
It was not an appetizing sight.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Though the empty shells of giant shipworms have long been common, exactly where these shells were coming from remained a great mystery to scientists (in spite of their name, it was not ships). The critical insight came when one of the project scientists discovered a documentary on Filipino TV showing the shells planted in the bed of a local shallow bay "like carrots". With this clue in hand, the scientists were at long last able to bag their quarry.
The scientists published their findings from their study of the worm in PNAS Early Edition this week, and they shed much light on an animal so rare that its biology, habitat, anatomy and even diet have largely been a mystery. The answers to these questions lend support, they says, to a bigger idea proposed nearly 20 years ago by one of the study's authors: the wooden steps hypothesis.
With a name like giant shipworm (Kuphus polythalamia), you would not be faulted for expecting this creature to live in ships. Although as we have seen, the giant shipworm eschews ships for submerged rotting wood and mud, the rest of the shipworms do live in ships. But all of the shipworms, the giant one included, are not actually worms. They are bivalve mollusks that have evolved to look like worms.
Bivalves are they of the hinged shells at the seafood buffet -- oysters, clams, scallops, cockles, and mussels. Somewhere along the line, a standard-issue bivalve decided to go in a different creative direction from its filter-feeding kin and set its sights on a new food source: sunken wood.
As long as there have been trees, there have been trees that wound up in the ocean. There they lay, appetizingly close but frustratingly out of gastronomic reach due to their content of polymers like cellulose and lignin. These chemicals – which stiffen wood -- have given hungry terrestrial creatures a run for their money too. They are famously indigestible to all but some fungi and bacteria. Animals who wish to eat wood must host microbes that can digest the tough fibers, as termites and beavers do.
Shipworms ultimately solved the problem in the same way: they allowed symbiotic bacteria who can digest wood to shack up in special cells in their gills. Then, in the process of boring into wood to consume it, a hinged shell containing a shellfish evolved into a tubular shell containing a worm.
Shipworm damage to driftwood. Credit: Michael C. Rygel Wikimedia (CC BY-SA 3.0)
Everyone was happy with this system until some hairless apes got the cockamamie idea to build their ocean-going vessels out of wood, whereupon the shipworms promptly chowed down, to the horror of the apes. This problem was not solved until the Royal Navy began sheathing their boats’ hulls with copper in the 18th century, which had the added advantage of discouraging pesky seaweed.
In any case, this blog post is not about those shipworms, but their wayward relative the giant shipworm, who once more split with the rest of the shipworms due to creative differences. Their new direction was inspired by some bacteria living nearby.
While shipworms were busy digesting all that lovely wood, the free-living bacteria around them were busy too. Some of them, in the process of digesting wood in environments devoid of oxygen, breathed the chemical sulfate, a component of seawater, instead. The gas they exhaled was hydrogen sulfide, the stinky chemical humans associate with rotten eggs. As it turns out, hydrogen sulfide can be used much like sunlight by another group of bacteria (the thioautotrophs). They can use the power contained in hydrogen sulfide to turn carbon dioxide into sugar. In other words, they can make their own food, just like plants.
At some point, these thioautotrophic bacteria invaded the gills of a shipworm living nearby. The scientists could tell this is the case because they put giant shipworm gills under the microscope. Inside the gills they found bacteria that contain sulfur globules and organelles called carboxysomes in which carbon dioxide can be turned into sugar. Both structures are common in thioautotorphic bacteria.
When they sequenced the bacterial DNA isolated from the gills, it closely resembled that of other thioautotrophic bacterial living inside marine microbes and hydrothermal vent snails and slugs. The sequence contained all the genes necessary to manufacture sugar from hydrogen sulfide.
Finally, they were able to isolate these bacteria in pure culture designed to grow thioautotrophic bacteria. The bacteria in this group are not closely related to the wood-digesting bacteria that other shipworms host. However, these formerly dominant bacteria were also present in the gills of the giant shipworms, though the evidence suggests they are rare – and highly disappointed.
As giant shipworms shifted their income stream from eating wood to hosting self-powering bacteria, their cecum, where bites of wood are stored; the organs in which shipworms digest food; and the muscles they use to bore into wood all atrophied. Only trace quantities of feces were found in the gut of the specimens these scientists digested, indicating that in addition to not needing to eat (because their in-house bacteria are doing all the cooking), these worms have, consequently, little need to poop.
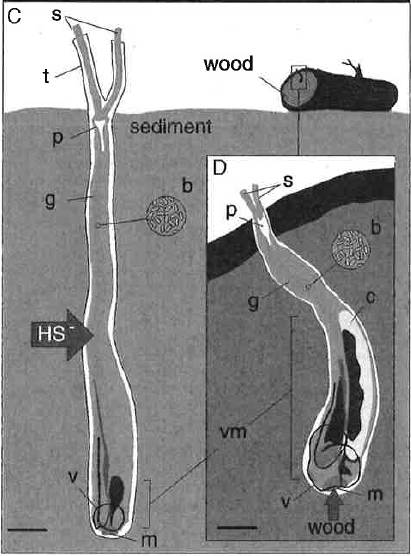
The giant shipworm (Kuphus polythalamia, left) and a wood-boring shipworm (Lyrodus pedicellatus, right). b=bacteria, c=cecum, g=gill, HS-=hydrogen sulfide, m=mouth, p=pallet, s=siphon, t=calcareous tube, v=valve (shell), vm=visceral mass. Scale bar on left, 5 cm, on right, 0.5 cm. Credit: Distel et al. 2017
Giant shipworms do have a mouth, but they’ve burned that bridge. They have walled their mouth off with their shell, and only periodically resorb it to extend their tubes. In place of all these shriveled organs, the bacteria-hosting gill has evolved to take up almost the entire worm. Based on this evidence, eating wood, sediment, or filtering water for food are unlikely to enable these worms to become giants, the authors conclude. Instead, growing fat and happy on the proceeds of their new bacterial brand partners, the worms grow into giants that commonly reach three feet and may reach five.
Interestingly, although becoming a worm is uncommon among bivalves, hosting thioautotrophic symbiotic bacteria is not. Five other families of bivalves do this, so perhaps the worms had some pre-adaptations that made this particular career change easier.
The scientists also argue that shipworm biology bolsters an old idea of lead author Daniel Distel: the “wooden-steps” hypothesis. This idea holds that the large thioautotrophic mussels that live around deep-sea hydrothermal vents evolved from smaller mussels that lived on sunken wood. These ancestor mussels used hydrogen sulfide produced by the bacteria that decay wood, but vent fluid was an easy substitute. Today, many bivalves and several tubeworms live equally well at hydrothermal vents, cold seeps (places where hydrogen sulfide, methane, and other hydrocarbons seep from the seabed), and in sunken wood. Wood is thus a stepping stone that can introduce thioautotrophic animals to hydrothermal vents and then help them spread between vents by island-hopping sunken wood like deep-sea Polynesians.
Now it looks as if the giant shipworm also had a layover in wood before arriving at its ultimate destination in the reeking sediments of the Philippines. Is it possible that the bivalves and the tube worms also began by eating wood, and only later stumbled on the culinary delights of hydrogen sulfide? If so, wood – a substance produced only in the glittering realm of the sun -- was not just a stepping stone between habitats in the deepest darkest ocean, but also between entire ways of life.
Reference
Daniel L. Distel, Marvin A. Al;tamia, Zhenjian Lin, J. Reuben Shipway, Andrew Han, Imeleda Forteza, Rowena Antemano, Ma. Gwen J. Penaflor Limbaco, Alison G. Tebo, Rande Dechavez, Julie Albano, Gary Rosenberg, Gisela Concepcion, Eric Schmidt, and Margo G. Haygood. "Discovery of chemoautotrophic symbiosis in the giant shipworm Kuphus polythalamia (Bivalvia: Teredinidae) extends wooden-steps theory." PNAS Early Edition. Published online before print April 17, 2017, doi: 10.1073/pnas.1620470114