This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American

It is the Age of Dinosaurs. Fighting apatosaurines by Brian Engh. Duck photographed by Darren Naish.
Fossil hominins, weird extinct lizards and archaic turtles are all very nice but, let’s be honest: when we talk about fossil tetrapods, the things we talk about the most are archosaurs... dinosaurs and their ilk in particular. As I’ve said on so many previous occasions, dinosaurs are always in the news, and while much of the stuff that gets classed as ‘newsy’ seems oh-so-familiar to those of us who attend the conferences and keep tabs on the technical literature, I know that isn’t the case for the Tet Zoo readership as a whole. That disclaimer out of the way, let’s look at some of the dinosaur-themed things that have lately been the topic of our attention.
A ‘mosaicy’ ceratopsian, called Mosaiceratops. The fossil record provides us with an incredible number of animals that possess weird combinations of features that are otherwise typical of distinct groups. What I’m trying to say is that there really are evolutionary ‘intermediates’ and – ugh – ‘missing links’ all over the place. One of the newest is a Chinese ceratopsian (the group conventionally termed the ‘horned dinosaurs’, even though they don’t all have horns) that lived about 85-90 million years ago, during the Late Cretaceous. Dubbed Mosaiceratops azumai, it possesses features typical of both neoceratopsians (the huge group that includes classic taxa like pig-sized Protoceratops and rhinocerine Triceratops), and of psittacosaurs (the deep-faced, bipedal ceratopsians, often imagined as ceratopsian prototypes relative to the more quadrupedal, bigger-headed neoceratopsians) (Zheng et al. 2015).
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
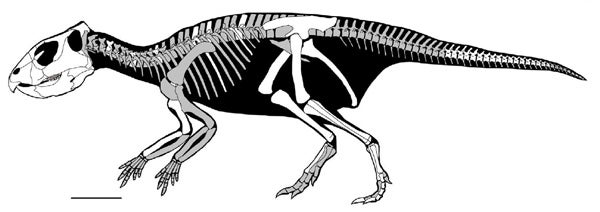
Skeleton reconstruction of Mosaiceratops, from Zheng et al. (2015). Scale bar = 10 cm.
Mosaiceratops is like a neoceratopsian in having a long predentary bone, a sloping upper margin to its snout, and a shortened post-dentary section of the lower jaw. But it’s like a psittacosaur in lacking premaxillary teeth, in having a big premaxillary bone and in having nostrils high on the side of the snout. In combining the features of both groups, it does make psittacosaurs (which are, otherwise, very weird compared to other ceratopsians) slightly less weird than they were before. Like other ceratopsians in this region of the cladogram, Mosaiceratops is small (about 1.3 m long) and with proportions suggesting bipedality.
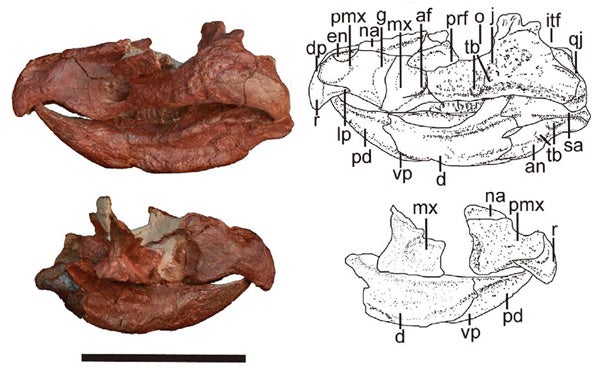
The partial skull of Mosaiceratops, from Zheng et al. (2015). Scale bar = 5 cm.
In phylogenetic analysis, it falls out as the sister taxon to all other neoceratopsians, other early-diverging members of this clade including North American Aquilops, and Asian Liaoceratops, Yamaceratops and Archaeoceratops. Notably, several of those taxa are several or many millions of years older (perhaps, in cases, as much as 30 million years older), so Mosaiceratops has a decent ghost lineage and close relatives (maybe other species) await discovery.
However... I must confess to being somewhat sceptical of the phylogenetic position proposed for this animal. Yes, some of its features do look somewhat psittacosaur-like, but they also look somewhat like those of a group of neoceratopsians not discussed by Zheng et al. (2015): namely, the bagaceratopsids (or bagaceratopids). These animals are deeply nested within Neoceratopsia, and hence well away from the location on the tree occupied by psittacosaurs, but they look similar to supposedly ‘psittacosaur-like’ Mosaiceratops (they lack premaxillary teeth, the nostril is dorsally located, the lateroventral wing of the premaxilla is large, and the predentary and mandible as a whole is similar to that of Mosaiceratops). Look at this montage...
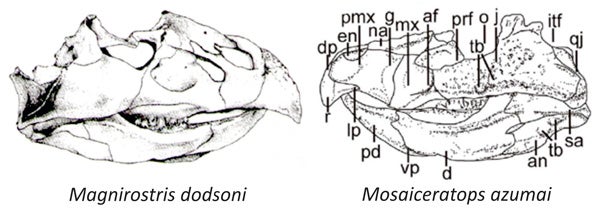
Magnirostris, a ‘bagaceratopsid’, from You & Dong (2003); Mosaiceratops from Zheng et al. (2015).
Then again, note that the lateroventral wing of the premaxilla in Magnirostris (a possible synonym of Bagaceratops) is not nearly as extensive as Mosaiceratops, plus Mosaiceratops has a much smaller rostral. On balance, this similarity doesn't sway me and I suspect that the authors are right. I’d welcome the thoughts of those who know ceratopsians better than I do.

The paired premaxillary fangs of some neoceratopsians - like Protoceratops andrewsi here - really are something quite special. What's the story here? Photo by Dave Hone.
Anyway, Zheng et al. (2015) make the interesting suggestion that the lack of premaxillary teeth in both psittacosaurs and Mosaiceratops indicates that their presence in other neoceratopsians is a novelty, not a feature inherited from early ceratopsians. Those premaxillary teeth – or fangs, if you like – sure are interesting, and we might wonder why certain neoceratopsians possessed them in the first place. There’s much more that could be said, but I have to stop there (and I haven’t even mentioned chaoyangsaurids).
Battling brontosaurs. For some years now, Mathew Wedel, Mike P. Taylor and I have been seriously discussing the idea that the insanely weird necks of apatosaurine sauropods might have been specialised intraspecific combat devices. Apatosaurinae is the diplodocoid group that includes Brontosaurus and its close kin (Taylor & Naish 2005) (and see this Tet Zoo article for a summary of the Brontosaurus situation), and their necks really are very weird compared to those of other sauropods.
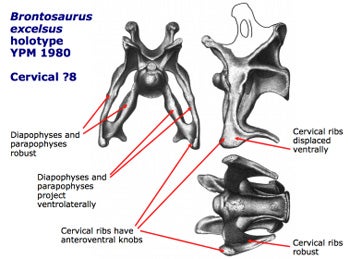
Several of the weird features of apatosaurine cervical vertebrae, as compiled on a slide by Mike Taylor. Image taken from here on SV-POW!
How weird? Their necks were broad and triangular in cross-section, not cylindrical. The individual vertebrae have massively constructed cervical ribs that are borne on thick, lateroventrally projecting (that’s outwards-and-downwards) beams, and the lower surfaces of the cervical ribs possess club-like bony bosses that might have supported enlarged keratinous extensions. There’s no indication that these features are especially relevant to a role in feeding or foraging, nor do they enhance the neck’s flexibility or anything like that. They really, really look like combat structures. Furthermore, they have analogues in the necks of certain modern animals that use their necks in fighting (more on that in future).
So – working together with artist and outstanding human Brian Engh – we’re seriously thinking that the unusual features of these necks evolved as combat adaptations. Brian has produced some kickass artwork that accompanies the whole project. And, no, this hypothesis is not at odds with our previously published contention that sauropod necks likely did not owe their weirdness to sexual selection pressure (Taylor et al. 2011). I’m deliberately being terse here since our research is not yet finished, our full case not yet put forward in print. Mike gave a talk about the hypothesis at a recent conference, and it’s been covered in several articles at the sauropod-themed blog SV-POW! Understandably, the whole idea has caught the zeitgeist and those of artistic bent have been falling over themselves to illustrate bull apatosaurines knocking the crap out of each other. This whole issue will be covered again in time, watch this space.

The web is now awash with images of battling apatosaurines. Here's one of our 'official' pieces, by Brian Engh. More on this subject in the future.
Neornithine family tree: MISSION ACCOMPLISHED... perhaps. Maybe 30 years ago, the shape of the bird family tree was described as an intractable mess, as an unresolved ‘last frontier’ where lawlessness and anarchy ruled, and a place where ‘trees’ were more ‘grass’ than anything else. Over the years, an absurd number of ideas have been put forward as goes the ways in which the different living bird groups (living birds = neornithines) might be related to one another. But since 2006 or so (Ericson et al. 2006, Hackett et al. 2008) a concerted effort from molecular biologists (sometimes aided and abetted by morphologists and even palaeontologists) has resulted in the emergence of a new ‘consensus’ tree for neornithines. A succession of studies, all incorporating and analysing an impressive quantity of genomic data, have recovered similar trees, though with various of the more problematic lineages appearing to be of yet-unresolved position (Suh et al. 2011, 2015, Yuri et al. 2013, Jarvis et al. 2014).
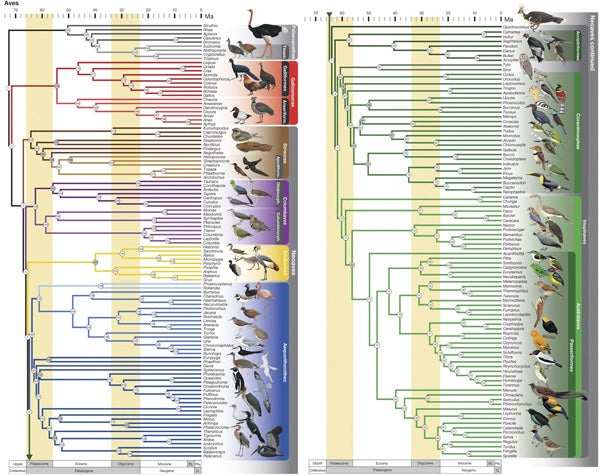
The Prum et al. (2015) bird phylogeny, beautiful illustrations depicting the groups concerned. See the paper for the full-sized version.
The latest such effort has just seen print (Prum et al. 2015) and it’s been getting some fanfare as the ‘most resolved’ of these studies so far. As is typical, this tree has confirmed several things we’ve long thought likely to be true. Example: a sister-group relationship between galliforms and anseriforms within Galloanserae, and a position for palaeognaths and galloanserines outside Neoaves, the grand clade that contains everything that isn’t a palaeognath or galloanserine.... At the same time, it gives us some seemingly weird ideas that definitely weren’t considered ‘consensus’ prior to a few years ago. Example: seriemas, falcons and parrots are close kin of passerines, bustards are nested within a clade composed of turacos and cuckoos termed Otidimorphae, and mesites, sandgrouse and pigeons form a clade termed Columbimorphae.

Molecular results consistently find bustards to group with cuckoos... that's so awesome. Great bustard Otis tarda (at left) by Andrej Chudý, CC BY-SA 2.0. White-browed coucal Centropus superciliosus by Adrian Valenzuela, CC BY-SA 2.0.
These findings aren’t novel to this study, but what is novel is the way in which various of these clades are combined. To stick with Otidimorphae and Columbimorphae, Prum et al. (2015) find them to be sister-groups (forming Columbaves), whereas previous studies found Columbimorphae and Otidimorphae to simply be somewhere in this approximate region of the tree, not specifically allied to one another, and perhaps with affinities to various of the other lineages present in this same approximate region (like Strisores/Caprimulgimorphae). All in all, Prum et al. (2015) provide a tidier, ‘lumpier’ tree where the many neoavian lineages fit within a low number of big assemblages.
Don’t get me wrong, I think this study is great, and I’m happy that we’re (apparently) seeing ever more resolved results. But there are a few things here that might not be as solid as they sound. Are clades like Columbaves really all that secure, given that it would take movement across (literally) one node for one of the constituent lineages to leave Columbaves and attach itself to a nearby clade? I mean, the whole point of certain other recent studies (Suh et al. 2015) was to show that rapid, explosive evolution and hybridisation at the root of several neoavian lineages meant that incomplete lineage sorting was a real thing early in neoavian history and that what we term hard polytomies – messy, star-shaped radiations – really did happen at some crucial early point in neoavian evolution. In other words, we might not be able to get precise resolution because precise resolution doesn’t exist. The ‘basal’ portions of the lineages concerned might have been messy, reticulated things.
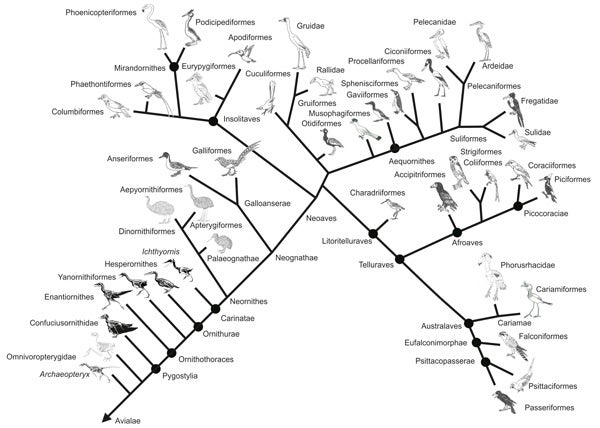
This old thing is looking a bit dated now... it's a schematic depiction of bird phylogeny (with some fossil groups depicted), based on Harshman (2007), Hackett et al. (2008) and Yuri et al. (2013). Modified from Naish (2014).
So, the main point is that, yes, we now have a well resolved view of neornithine phylogeny. The majority of lineages consistently appear in the same approximate position in the trees generated by different studies. But does this new study necessarily represent ‘better’ resolution than what we’ve seen in other recent studies (Jarvis et al. 2014)? I’m not so sure. And I doubt that they’re right about tinamous being especially close to cassowaries and emus, but we’ll see.
For previous Tet Zoo articles relevant to various of the issues covered here, see...
Ryan et al.'s New Perspectives on Horned Dinosaurs: a review
Getting a major chapter on birds ALL birds into a major book on dinosaurs
Dinosaurs and their exaggerated structures: species recognition aids, or sexual display devices?
Ref - -
Naish, D. 2014. The fossil record of bird behaviour. Journal of Zoology 292, 268-280.
Prum, R. O., Berv, J. S., Dornburg, A., Field, D. J., Townsend, J. P., Moriarty Lemmon, E. & Lemmon, A. R. 2015 A comprehensive phylogeny of birds (Aves) using targeted next-generation DNA sequencing. Nature doi:10.1038/nature15697
You, H. & Dong, Z. 2003. A new protoceratopsid (Dinosauria: Neoceratopsia) from the Late Cretaceous of Inner Mongolia, China. Acta Geologica Sinica 77, 299-303.
Zheng, W., Jin, X. & Xu, X. 2015. A psittacosaurid-like basal neoceratopsian from the Upper Cretaceous of central China and its implications for basal ceratopsian evolution. Scientific Reports 5:14190. DOI: 10.1038/srep14190