This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
In 2003, the Mediterranean coral Oculina patagonica did something that was supposed to be impossible: it destroyed bacteria that had formerly been bleaching and killing it. This was a great shock, because the prevailing wisdom is that corals do not possess immune systems like ourselves, and thus have no ability to develop immunity. And yet, somehow, this coral behaved as if it had.
Vertebrate immune systems learn to recognize and remember many pathogens, so that when they attack again, we are “immune”. This is the principle on which vaccines work, and is why it is so fabulous that they save us the potentially lethal and decidedly unpleasant experience of suffering disease yet still make us immune as if we had.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Corals, on the other hand, enjoy no such privilege. Corals possess “innate” immunity – they can defend themselves with general responses like physical barriers, mucus, bacteria-eating cells, and chemical defenses. But they possess no customizable system of “memory” cells that learn what pathogens look like and sound the alarm early should they see them again.
Yet the disease-causing bacteria Vibrio shiloi – previously proved to cause the disease in this species by rigorous application of Koch's postulates – were no longer multiplying wildly in the coral as they had done. Instead, they invaded and died within a few days.
What could have happened? Eugene Rosenberg and Ilana Zilber-Rosenberg had an idea: what if the coral's well-known menagerie of in-house symbiotic bacteria had acquired a new member with some bacterial butt-kicking powers? Or perhaps one of the existing crew had mutated in a way that now enabled it to kill the invading bacteria? Or perhaps one of the symbionts had a fling with a passing bacterium that gave it a resistance gene vital to destroying Vibrio shiloi?
Bacteria can evolve and adapt much faster than a stony coral can. What if the symbionts were behaving as a de facto immune system for the coral? They dubbed this idea the “Coral probiotic hypothesis”, and soon extrapolated the idea to encompass all symbiotic creatures in the “Hologenome Theory of Evolution”. This postulates that the true unit of natural selection is not a single organism, but the collective of an organism and all the symbiotic micoorganisms living with it.
Like many new ideas, the theory has attracted its share of criticism and praise. But a possible new example of the phenomenon recently emerged in another organism well-known for its deeply symbiotic lifestyle: lichens. And in this case, a change of symbiont may have had a different, but no less radical effect on the biology and evolution of the organism, as documented in a review article by a team of German and Spanish scientists published in Nature Reviews back in 2007.
Lichens are co-ops composed of fungi, algae, and, as recently realized, bacteria and archaea. The fungus builds the “house”, and the algae lives sandwiched inside. Though often enshrined as the model of a happy symbiosis, the extent to which the algae benefits in any given scenario varies. The fungus, in particular, seems to like to snack on it.
In this case, our hero is the humble dark brown lichen Cetraria aculeata, which has an enormous range encompassing both the sunny Mediterranean and the frosty Antarctic. It lives in Arctic and Antarctic heath as well as coastal sand dunes, woodlands, and steppes in temperate and dry regions of the Mediterranean, central Asia, and Patagonia.

Cetraria aculeata in its happy place. Fig. 1 from Printzen et al. 2007.
It turns out this state of affairs is not uncommon – many lichens boast both enormous ranges and wide ecological niches. But it is rare in other organisms. Most polar creatures are intimately bound to their environment and can live there and nowhere else. The phenomenon may be due to lichens' ability (which they share with mosses and some algae) to put themselves into suspended animation when they dry out – an ability called poikilohydry. Most plants cannot do this and wilt and die under similar circumstances. Mosses and lichens shrivel – but do not die. When touched by water again, they quickly revive and begin the business of life again.
But this cannot be the whole story. To survive and photosynthesize in extreme cold and wild swings in light and temperature surely requires adaptations that go beyond a simple ability to hibernate. It could be that we have defined lichen species too broadly, and there are “cryptic” species with smaller ranges hidden within the species as defined today. It is also possible that lichen-forming fungi possess extraordinary powers of acclimation. And there is another possibility.
For a symbiotic organism, one way to quickly acquire a new habitat without the bother of slowly adapting to it may be to simply shanghai a new symbiont. Lichens are well known for their ability to play host to multiple symbiotic algae, sometimes at the same time. Many lichens even make special houses called cephalodia for their blue-green bacterial symbionts to keep them separate from their plant-like eukaryotic green algal symbionts (do they not get along?)

"PeltigeraAphtosa20090629" by Tigerente - Own work. Licensed under CC BY-SA 3.0 via Wikimedia Commons. The dark gray freckles on this freckle pelt lichen (Peltigera aphthosa) are the cephalodia in which it houses its blue-green bacterial symbionts. The green algal symbionts are distributed thoughout the rest of the lichen and give it its grassy color.
It has long been known lichens can switch algal partners with ease. The lichen fungus sometimes switches during different parts of its life cycle, but they may also associate with different algae in different parts of their geographic range.
There is evidence this is exactly what happened in C. aculeata. Mediterranean populations of both symbionts are genetically isolated (in the case of the alga, 100% isolated)– that is, they've stopped exchanging genes, meaning they no longer interbreed -- from polar populations. This suggests “a historic [algal] switch enabled C. aculeata to colonise its wide ecological niche,” the authors write.
Although there's no evidence right now that lichens swap their algae quickly and repeatedly, such switches would allow the lichen-making fungus to react much more quickly to environmental change than the glacial pace of mutation and selection in long-lived lichens, they say. It could allow them to quickly conquer new niches and it may even be a major method by which lichens speciate. And it could help explain the gargantuan home ranges of many lichens.
The authors are not certain, however, in which direction the switch happened. The genetic diversity of both host and symbionts are higher in the Arctic. That may suggest the Arctic is the true homeland as species established in a place for a long time tend to show the most genetic diversity, while recent arrivals tend to be more genetically uniform. That is the case in Antarctica, where the lichens are almost identical to each other. On the other hand, family trees constructed by comparing lichen genes suggest that the Mediterranean may be the true birthplace of the lichen, and it was the Arctic that was conquered by a partner swap.
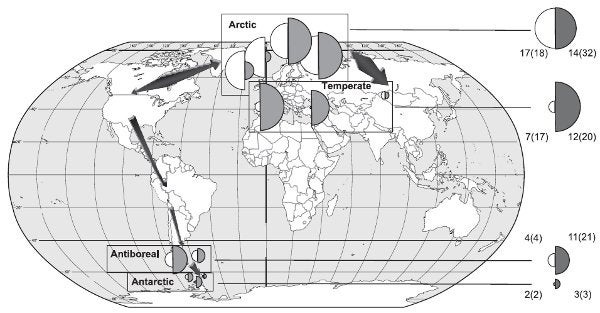
Genetic diversity of different populations of the lichen fungi Cetraria aculeata (white) and their algal partners (grey). "Size of the semi-circles is proportional to the diversity level. Circles on the right summarize diversity levels within regions." Fig. 4 from Printzen et al. 2007.
Scientists who have considered this subject agree that symbiosis likely increases the adaptive and evolutionary potential of the partners by allowing for quick evolutionary pivoting in organisms that on their own are by nature staid. “A symbiotic host may adapt to changing environmental conditions by “outsourcing” parts of its stress response to the symbiotic partners,” the authors write.
In effect, slow-evolving organisms like lichen-forming fungi and stony corals (both of which are famously long lived, on the order of centuries or even millennia) may make themselves more evolutionarily nimble – and even MacGyver an immune system or conquer the Arctic in a single bound – by swapping symbionts like parts on a Mr. Potato Head.
References
Printzen, C., Domaschke, S., Fernández-Mendoza, F., & Pérez-Ortega, S. (2013). Biogeography and ecology of Cetraria aculeata, a widely distributed lichen with a bipolar distribution. MycoKeys, 6, 33-53.
Rosenberg, E., Koren, O., Reshef, L., Efrony, R., & Zilber-Rosenberg, I. (2007). The role of microorganisms in coral health, disease and evolution. Nature Reviews Microbiology, 5(5), 355-362.