This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
The typical bacterial to-do list is not long. Eat. Grow. Divide. Repeat. But there's one more important item on that list: avoid Bdellovibrio.
Bdellovibrio bacteriovorus's job is to ambush and kill other bacteria, and it approaches that calling with verve. After torpedoing toward its prey, it drills a porthole not quite as wide as itself, squeezes into the space between the cell wall and the inner membrane, and then plugs the opening. There, the predator steals food and grows like a demonic sausage before dividing into links of equally ill-intentioned offspring that burst from their victim’s corpse.
The entire process takes only four hours, but the prey is dead within 20 minutes. The predator keeps its victim's body intact for another four hours, however, to permit what the authors of a new study on Bdellovibrio coolly refer to as “private dining”.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Though bacteria-on-bacteria predation like this is likely common, scientists have spent a lot more time studying viruses, protists, and animals that kill and eat bacteria, and the extent to which bacterial predators of bacteria are important remains fuzzy.
What is known is that predatory bacteria, while not abundant (what predators are?), are widespread in nature. They’ve been detected in soil, rivers, the ocean, water treatment plants, crab gills, oyster shells, chicken s***, and even human waste. Which means there could be some inside you right now (Cool!).
They're also fast. Due to a turbo-charged sheathed propeller called a flagellum, they can achieve velocities of 100 body lengths-per-second, making them the current bacterial land-speed record holders – the cheetahs of the bacterial Serengeti (actual cheetahs make 16 body lengths/second).
Recently, scientists were curious to what extent B. bacteriovorus directs the remodeling of its victims’ walls to suit its own designs.
Bacterial cell walls are built of peptidoglycan, a mesh of short chains of amino acids – the building blocks of proteins -- called oligopeptides, and chains of sugars called NAG and NAM.

Credit: Mouagip Wikimedia
Peptidoglcyan forms a cage around the bacterial cell; the authors of this study call it a “shape-determining” macromolecule, which is a fantastic euphemism. Peptidoglycan: slimming and body sculpting! The Spanx of biochemicals, really.
In this respect, it’s not much different from the chitin used by fungi or the cellulose and lignin used by plants to corset and constrain themselves, to sculpt their cells and keep them from bursting when inundated by water.
Lignin, a biochemical work of art. Seriously, you could frame this sucker and hang it in a gallery. Anytime you touch wood, you are touching something like this. Credit: Karol Głąb Wikimedia (CC BY-SA 3.0)
When enzymes deployed by B. bacteriovorus replace the original amino acids in the crosslinks of peptidoglycan with different amino acids – fluorescently labaled by scientists in this case, but also available in nature -- the replacements strengthen the peptdioglycan mesh.
This chemical reinforcement is not, the authors say, unlike the art of fresco. A frescoist applies pigments to wet plaster such that they become impregnated in the wall itself as the plaster dries. This is how da Vinci created The Last Supper and Michaelangelo painted the Sistine Chapel. In similar fashion, the amino acids swapped by Bdellovibrio in the peptidoglycan mesh bond to it, but rather than add color, they add strength.
When scientists previously studied Bdellovibrio with electron microscopes, their images suggested the sequence of predatory events outlined at the top of this post. But because the cell walls of both predator and prey are biochemically similar, it was hard to be sure exactly what was going on, particularly where the two walls met.
To that end, this team employed fluorescently-tagged wall-fortifying amino acids to see exactly what was being removed or added by the predator. When they placed the samples under a three-dimensional fluorescence microscope, this team of American, British, and French scientists were able to verify these earlier hypotheses were more or less correct, and published their results in Nature Microbiology last year.
In the images below, Bdellovbrio's cell wall glows red while newly-added wall-strengthening construction materials glow blue. Around the time B. bacteriovorus entered its prey, the microscope revealed a blue ring just slightly smaller (0.24 micrometers) than the predator’s diameter (0.3 micrometers ), evidence of both the hole and its reinforcement. The arrowheads point out the rings below.
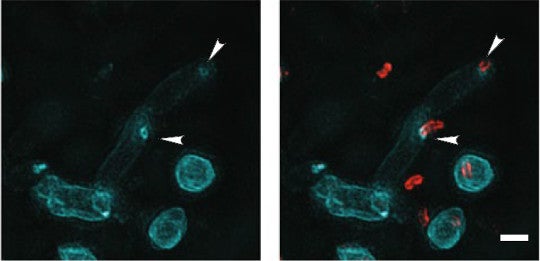
Credit: Kuru et al. 2017
Bdellovibrio carries on reinforcing the prey cell wall generally, not just around the hole. Whether as a result or via a different mechanism, the host cell also becomes round. You can both phenomena above, lower right.
In some cells, the wall also warped around the hole, perhaps evidence of the action of the predator’s wall-boring enzymes and of the need for chemical reinforcement.
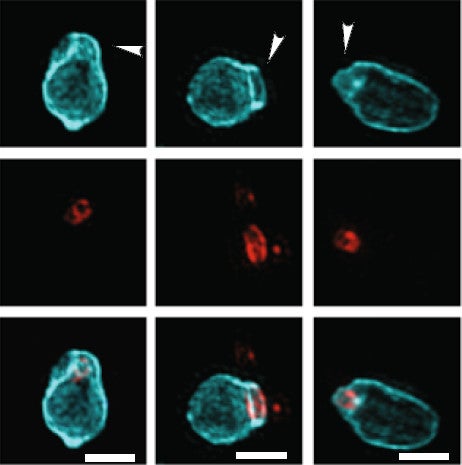
Credit: Kuru et al. 2017
Later, in a small percentage of prey cells, a blue disc appeared near the entry point.
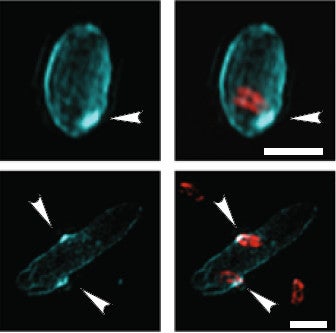
Credit: Kuru et al. 2017
This may be a plug constructed by the predator to seal itself in to its VIP all-you-can-eat buffet.
The system this bacterium has devised for feeding and reproducing at its own leisure is not unlike a big cat that drags a fresh kill into a tree so it can eat in peace. Except this predator is smaller, faster, and doesn’t sleep 12 hours a day. Consider yourselves lucky, gazelles.
References
Erkin Kuru, Carey Lambert, Jonathan Rittichier, Rob Till, Adrien Ducret, Adeline Derouaux, Joe Gray, Jacob Biboy, Waldemar Vollmer, Michael VanNieuwenhze, Yves V. Brun, R. Elizabeth Sockett. "Fluorescent D-amino-acids reveal bi-cellular cell wall modifications important for Bdellovibrio bacteriovorus predation." Nature Microbiology, 2017; DOI: 10.1038/s41564-017-0029-y
Yair, Shemesh, Davidov Yaacov, Koval Susan, and Edouard Jurkevitch. "Small eats big: ecology and diversity of Bdellovibrio and like organisms, and their dynamics in predator-prey interactions." Agronomie 23, no. 5-6 (2003): 433-439.