This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
Plankton, it turns out, are not just floating happy meals. The marine microbes are actually armed and dangerous, equipped with weapons capable of firing filaments or mucus to foil the guns of their enemies in an evolutionary arms race-cum-naval battle.
And with good cause: the guns of their microbial predators, we are learning, are numerous, fearsome, and more sophisticated than we had ever imagined.
Observe: the microscopic Wild Kingdom.
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
Below, next to the white arrows, you can see the ballistic defensive hairs fired by prey in response to attack.
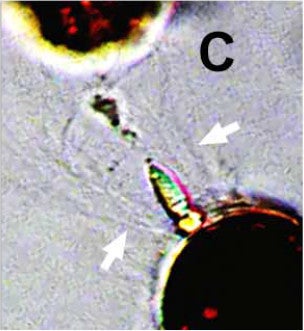
Credit: Gavelis et al. 2017
Scientists from Canada, Japan, and Germany decided to take a closer look at some of those weapons. They examined the ballistic guns of predatory marine microbes called dinoflagellates, the ones you have just seen, due in part to their tantalizing resemblance to those found in stinging animals like coral and jellyfish.
They were startled by what they found, publishing it in Science Advances last March. The microbial versions surpass the animals’ in complexity and seem to have been invented independently. On top of that, dinoflagellates seem to have stumbled upon the Gatling gun -- or something very like it -- millennia before Richard Gatling.
Dinoflagellates are protists, microscopic life forms that contain a DNA-packed nucleus and other specialized cellular machinery. Their sometimes elaborately sculpted shells bear two propulsive tails, and, often, a certain punk aesthetic. Many dinoflagellates make their own food from light. Others prey on other microbes, and some hedge their bets by doing both. A protist’s gotta do what a protist’s gotta do.
For this study, the scientists chose to focus on the dinoflagellate Polykrikos, the organism you saw in the movie nonchalantly hauling its prey off for dinner. Its dramatically sculpted shell gives the appearance of a curl of carefully piped frosting.

Credit: Gavelis et al. 2017
Tucked into the fold of the curl are organelles called nematocysts. These lidded torpedo tubes contain a needle-tipped cable that explosively punctures prey when the chamber is triggered by touch.
At first, scientists suspected that nematocysts found in dinoflagellates evolved from a weapon first created by a shared common ancestor of Cnidarians, the supergroup of animals to which jellyfish belong. But dinoflagellates and cnidarians are actually very distantly related. So that seems unlikely.
On the other hand, some photosynthetic dinoflagellates live symbiotically inside the polyps of coral, another cnidarian. Such physical intimacy might have provided the necessary ambiance for a nematocyst donation.
They decided to take a closer look.
When they scrutinized the proteins involved in building and firing the nematocysts, they discovered that the nematocysts in dinoflagellates and cnidarians were not closely related (although the protein that generates the nematocyst propellant in jellyfish does seem to have been “borrowed” from bacteria). Most of the genes in cnidarian nematocysts appear to have evolved independently after the appearance of animals.
Similarly, the proteins that build the dinoflagellate nematocyst bore no similarity to those that build similar weapons in other predatory marine microbes like ciliates or
cryptophytes. If that’s the case, then the striking similarities between dinoflagellate and all other nematocysts are simply a colossal coincidence -- and yet another example of the wildly common phenomenon of convergent evolution. Similar evolutionary pressures seem to generate the same solutions on this planet over and over again.
Next they decided to compare the equipment itself. They used ion beam scanning electron microscopy to construct three-dimensional images of Polykrikos kofoidii.
They discovered differences that, though subtle, were significant. Each nematocyst was tethered to another slender organelle called a taeniocyst not found in cnidarian nematocysts. You can see it poking out of the dinoflagellate in the electron micrograph above, and here at top after the organelles have been dissected.
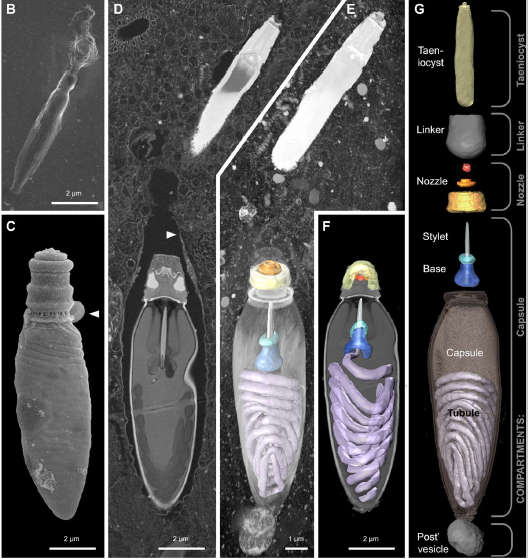
B)shows the mysterious contents of the taeniocyst discharged at top. White arrow in C) shows the popped off lid of a discharged nematocyst. White arrow in D) shows a "membranous chute" that encloses nematocyst. Credit: Gavelis et al. 2017
The lid on Polykrikos’s nematocyst overlays a complicated, three-ringed structure not found in cnidarians that the scientists termed a “nozzle”. The nozzle sits in a subcompartment of the capsule, which although surmounted by the lid, has no opening itself.
On firing, the stylet that caps the long tube coiled in the nematocyst punctures the capsule, pops off the lid, and then – if everything goes according to plan -- penetrates armored prey.
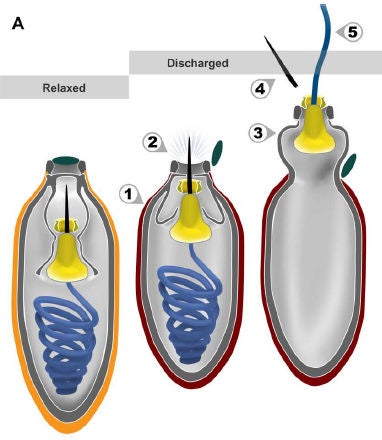
Hypothetical firing sequence. Relaxed (orange) capsule contracts (red), internal pressure forces stylet to pierce capsule and open operculum, part of capsule everts, stylet detaches, and tubule everts. In this concept, stylet is only involved in puncturing capsule, not prey. Credit: Gavelis et al. 2017
Cnidarian nematocysts do not need to be punctured to rupture. Their lid simply pops off.
To the scientists’ surprise and contrary to their expectations, this particular cable does not do the towing. It dissolves soon after deployment. Rather, its function seems to be to stab prey like an arrow or bullet. Ideally, this causes bleeding or perhaps a lethal influx of seawater, incapacitating and mortally wounding prey. A separate system – oddly located, in my opinion, on the other side of the dinoflagellate -- launches the tow cable.
The cigar-shaped taeniocyst poses a greater mystery. It contacts prey before the nematocyst and sits atop the nematocyst like the final stage of an Apollo rocket, connected to it by a newly discovered “linker” organelle.
Based on movies recorded by this team, the taeniocyst appears to be a ballistic organelle itself, explosively firing … something.
The scientists propose that the taeniocyst is the first stage of a three-part assault.
On contact with prey, the taeniocsyt launches something that adheres to the target. The nematocyst then fires, stabbing the victim, and finally the tow cable launches, allowing the predator to haul off its prize for consumption at its own convenience.
Attempting to understand how such a system evolved, the scientists decided to examine another dinoflagellate whose weaponry was known to be substantially different -- and even more intricate.
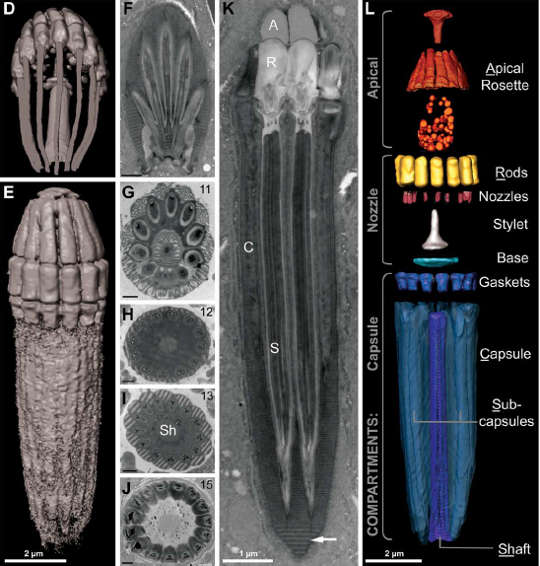
Nematodinium possesses no coiled ballistic tube or taeniocyst. What it has instead is a circular array of guns so profoundly different from what Polykrikos possesses that some questioned whether they shared an evolutionary origin. “… [E]ach nematocyst,” the authors write, “is essentially a bouquet of smaller ballistic organelles.”
Credit: Gavelis et al. 2017
This particular feat of engineering recalls one of the many innovative lethal inventions of the U.S. Civil War: the Gatling Gun.

Credit: Pearson Scott Foresman Wikimedia
The rotary gun was conceived as a way to enable more rapid fire by giving the barrels time to cool between firing.
In the hands of Nematodinium, however, the design functions differently. Instead of an operculum, the business end of the nematocyst is covered by what the scientists describe as “an elegant rosette” (see above). Instead of one nozzle, it contains a ring of nozzles sitting atop each subcapsule – but then again, only a single piercing stylet, just as in Polykrikos. At the base is a ring of 11-15 subcapsule barrels.
Once fired, the subcapsules extrude (or simply are) some sort of mucus-like material up to twice their former length. It does not appear that the nematocyst of Nematodinium actually fires like a Gatling gun, rotating, with only one barrel active at a time.
How do dinoflagellate nematocysts fire? They don’t use the same chemical propellant that cnidarians do. We know that. They may have a propellant of their own that we have yet to discover. However, their anatomy suggests another intriguing possibility. The capsules of both Polykrikos and Nematodinium both have grooved walls. It's possible, the scientists suggested, that the capsule wall contains contractile fibers. When activated, they collapse the capsule along its grooves, pressurizing it.
Some evidence for this exists. In the nematocysts seen under microscopy, the ridges were spaced at two different intervals. The more distant spacing may be the unfired state, and the other the fired.
As both organisms are in the same dinoflagellate kingroup, this further suggests, the scientists say, that in spite of the physical dissimilarity between the artillery pieces of Polykrikos and Nematodinium that they did, in fact, evolve from a common ancestor.
Here is one hypothetical scenario:
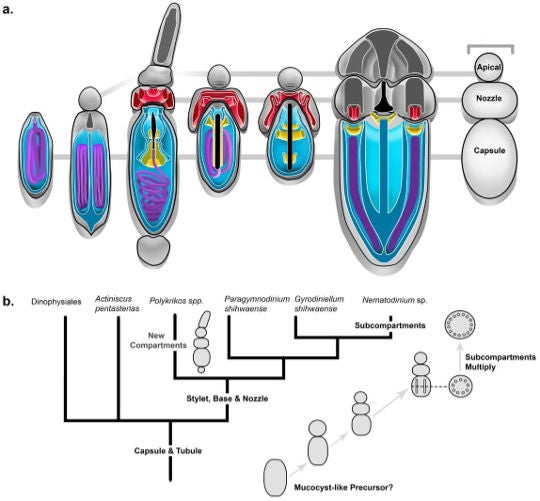
Credit: Gavelis et al. 2017
The ridges in the capsule are also yet another of the differences between nematocysts made by cnidarians and those made by dinoflagellates, summed up thusly for those with interest:
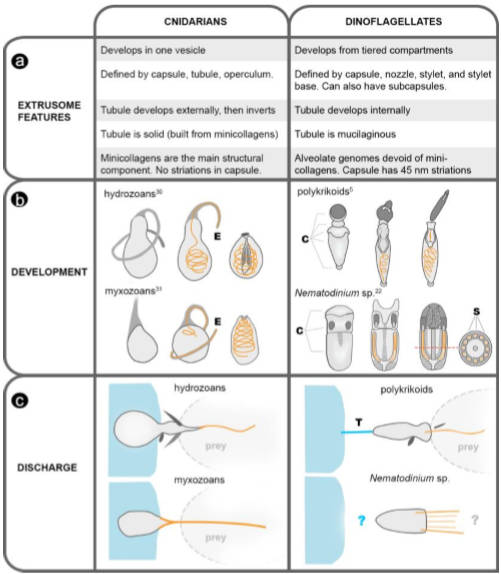
Fig S1 from Credit: Gavelis et al. 2017
The complexity of this microbial arsenal hints at the deadly seriousness of the evolutionary arms race among marine microbes. Such sophisticated and costly engineering as shell-piercing stylets, toxin-injecting tubules, and explosive propulsion systems seems likely to be a response, the scientists hypothesize, to the armor and defensive ballistics evolved by prey. In a previous study, these scientists found prey dinoflagellates – complete with their own fired, but apparently futile defensive weapons – inside wild-caught Nematodinium.
Indeed, these battles must occur continually. Up to 60% of the “primary production” of the ocean -- mostly biomass generated by marine algae, I would think -- is consumed by ciliates and dinoflagellates. Not blue whales.
Life has apparently come up with many, many ways to construct these intricate weapons. The pressure to attack and defend in the vast liquid free-for-all of the ocean must be intense. More than any other habitat, it’s the Wild West. And you wouldn’t dream of stepping onto Main Street at high noon without packing heat.
Reference
Gregory S. Gavelis et al. Microbial arms race: Ballistic “nematocysts” in dinoflagellates represent a new extreme in organelle complexity. Science Advances, 2017 DOI: 10.1126/sciadv.1602552