This article was published in Scientific American’s former blog network and reflects the views of the author, not necessarily those of Scientific American
Beams made of sawn diatom shells and poked with a diamond-tipped probe until they cracked have revealed that the microbial armor has the highest strength-to-weight ratio of any known biological material. According to the scientists who conducted the study, the remarkable toughness of this material is likely due to its honeycomb-like architecture and flawless silica build.
Diatoms are single-celled phytoplankton, or marine algae. They come in a dizzying array of shapes and colors much beloved by this author.

By Wipeter - Own work, CC BY-SA 3.0, https://commons.wikimedia.org/w/index.php?curid=5682386
On supporting science journalism
If you're enjoying this article, consider supporting our award-winning journalism by subscribing. By purchasing a subscription you are helping to ensure the future of impactful stories about the discoveries and ideas shaping our world today.
The scientists studied the shells – called frustules – of Coscinodiscus, a hamburger-shaped diatom. Like all diatoms, it bears two frustules that fit together like the halves of a Petri Dish. The shells are perforated by pores. Here's a view from overhead:

By Geographer - Own work, CC BY-SA 3.0, https://en.wikipedia.org/w/index.php?curid=43213425
And here's a closer look at the microstructure:
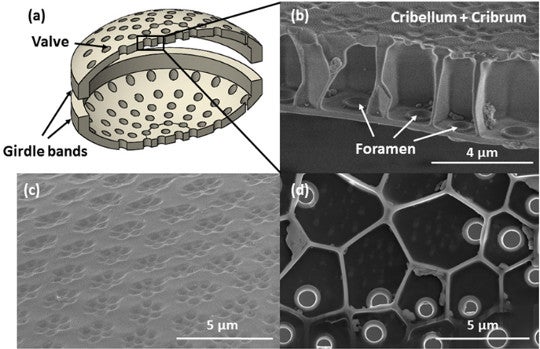
(A) Schematic of shell (B) Cross-section of shell showing honeycombing and rimmed frustules (C) Outer surface of shell, showing hexagons of pores arranged in meta-hexagons (D) Inner layer of shell, showing one foramen per cell. Fig. 1 from Aitken et al. 2016.
Smaller elliptical pores pierce the exterior of the shell in groups of six, the groups themselves arranged in hexes. Lining the frustule interior are larger holes called foramina (sing. foramen) ringed with reinforced rims. In between the inner and outer walls lie honey-comb shaped cells with five or six side walls, each perforated by one foramen. All parts of the shell are made of smooth and apparently flawless amorphous silica – i.e., glass.
Diatom shells serve many purposes: portals for nutrients, buoyancy control, shaping water flow around the shell, and, of course, protection from predators and viruses. All of these forces compete to sculpt the structure of the shell. Diatom shells grown in more predatory environments tend to be more fortified. On the other hand, a denser shell also may cause the diatom to sink to waters with too dark for photosynthesis and impair or prevent the exchange of gases, food, and waste that is as essential for diatoms as it is for us. Diatom shells have pores that may serve these purposes, but what has been unclear until now is to what extent those pores contribute to the strength of the shell, the need for exchange with the environment, or both. This team of scientists set out to test their influence – along with the rest of the shell’s structure -- on strength.
As you can imagine, conducting strength tests on structures measured in micrometers is a delicate business. Traditional mechanical testing methods cannot be used, and the tests conducted so far have given widely varying results. That’s perhaps because scientists performed these tests on whole shells, with all their irregularities.
In this experiment, scientists from Cal Tech and the Norwegian University of Science and Technology resolved some of the problems caused by testing slippery and bumpy shells by fabricating their own diatom beams – a feat almost as extraordinary to me as the discovery of the beam’s strength. They used a focused ion beam inside a scanning electron microscope to cut a plank from a diatom frustule on three sides and then affixed a tungsten needle to the beam with a bead of platinum. Then they milled away the fourth side and then lifted it out of place with a tungsten needle. They could then deposit it where they wished. In the case of the three point bending experiment, that place was suspended over a well 20 x 20 x 40 micrometers in size. The finished beams look just like a roof or wall beam, but made of diatom, and very, very tiny.

How a diatom beam is born, and the well over which it was placed for strength testing. Fig. S4 (rearranged) from Aitken et al. 2016.
They then tested these beams with the same experiment one would use on a beam of concrete or steel – a three-point bending experiment. Here’s one done at our scale:

By Rama - Own work, CC BY-SA 2.0 fr, https://commons.wikimedia.org/w/index.php?curid=863335
In this experiment, the scientists tested their beam in a scanning electron microscope armed with a “nanoindenter”, i.e., a tiny probe.

Three-point bending experiment on beam from diatom frustule. The beam is turned so the inside of the shell is facing us. Fig. 3A from Aitken et al. 2016.
The nanoindenter pushed down on the beam with steadily increasing pressure. When the beam finally snapped, it launched itself into the air so quickly it exceeded the image scan rate of the SEM – so the break couldn’t actually be seen. Most snapped samples simply vanished into the (relatively) cavernous space around them.
The scientists did manage to find one half of one cracked beam and take a picture of it under the SEM to see exactly how it broke and where. The pores in the outer surface of the diatom shell seemed to attract the fracture – or at least, not bar its path -- while the crack on the other side of the beam travelled conspicuously around a foramen.

(a) path of a crack shown on one side of a sample and (b) the path on the other side, extrapolated from the 1/2 beam that was recovered after fracture (insets, shown from two perspectives). Fig. 4 from Aitken et al. 2016.
As a result, the authors thus suggest the foramens in the basal plate fortify the shell, while the pores in the outer surface serve to channel resources.
Based on measurements of the average failure stress and strain, and of the samples’ density, the scientists were able to calculate the beams’ specific strength, or strength-to-weight ratio. The value -- 1702 kNm/kg -- is “well above” the values for other natural materials including bamboo, mollusk shell, and spider silk.
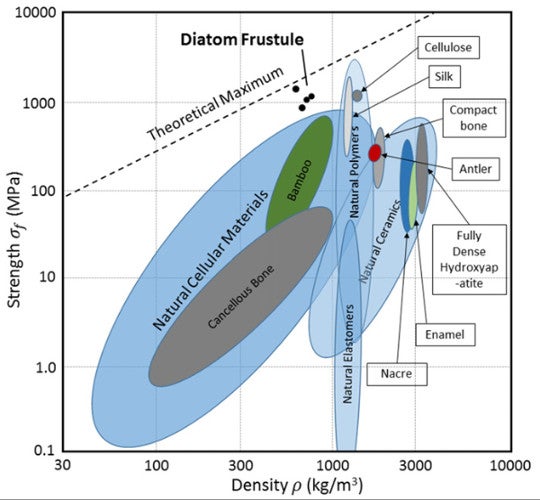
Strength-to-density ratios of natural materials. Fig. 5 from Aitken et al. 2016.
Diatom frustule is thus positioned in new real estate on the biologic materials strength-to-weight chart, achieving strengths comparable to silk and other natural polymers, but at lower density. The scientists attributed this feat to their honeycombed interiors and the flawlessness of their silica – no flaws were detectable down to a resolution of two nanometers.
That’s not to say no known substances exceed this specific strength – many do, but they are all man-made. Kevlar is one such substance, and another is high-purity silica nanowire, made, as implied, of pure silica. Diatom shells, by contrast, are actually composites of silica and an organic scaffolding used to construct them. For example, Coscinodiscus seems to use long-chain polymers as to “flocculate” (assemble) silica particles during shell fabrication.
These organic scaffolds seem to remain in place inside the shell walls after project completion. The cleaning procedure scientists used to remove the contents of the cell does not purge them, so they are likely chemically bound to the silica. Their staying power is testified to by the fact that such polymers have been recovered from the walls of diatoms extracted from Southern Ocean sediment cores nearly 8,000 years old.
Still, the bond between the scaffolding polymer and the silica has been hypothesized to consist of hydrogen bonds, which as every high school chemistry student knows, are weak relative to covalent bonds – the type that links the silica. So it is likely along the hydrogen-bond boundary between the silica and its organic scaffolding that diatom shells crack when placed under pressure – a “flaw” that silica nanowires lack.
Reference
Aitken, Zachary H., Shi Luo, Stephanie N. Reynolds, Christian Thaulow, and Julia R. Greer. "Microstructure provides insights into evolutionary design and resilience of Coscinodiscus sp. frustule." Proceedings of the National Academy of Sciences 113, no. 8 (2016): 2017-2022.